Guideline/version 556/04.02.00.01/2024/6
1 Introduction
1.1 Validity of the instructions
The Food Supplement Guide is intended for use by both food control authorities and food business operators. The purpose of this Guide is to provide a wide range of information on food supplement legislation, its application and control.
Public authority action shall be based on legislative competence conferred to the authority and be consistent with legislation. Regulatory guidelines are not, by their legal nature, binding on other authorities or operators. Issues pertaining to the application of legislative regulations are in the last instance settled by a court of law.
This Guide contains both direct citations from legislation and interpretations on the application of legislation. The legal interpretations presented in this Guide are views held by the Finnish Food Authority regarding how legislation should be applied. The Finnish Food Authority’s position may change if a different interpretation is made at the Community level, essential additional information is obtained or regulations change.
1.2 Legislation and guidelines
Legislation and guidelines on food supplements have been compiled on the Finnish Food Authority website. Other guidelines and legislation concerning the food sector can also be found on the same website. The latest consolidated version of the legislation must always be used.
A number of different regulations apply to food supplements, as with other foods (Figure 1). Food supplements also have several interfaces with other foods as well as with medicines and medical devices. It is important to take this into account both in operator own-check and control conducted by authorities. Consequently, this Guide also provides general information on other regulations applicable to food supplements and food supplement interfaces.
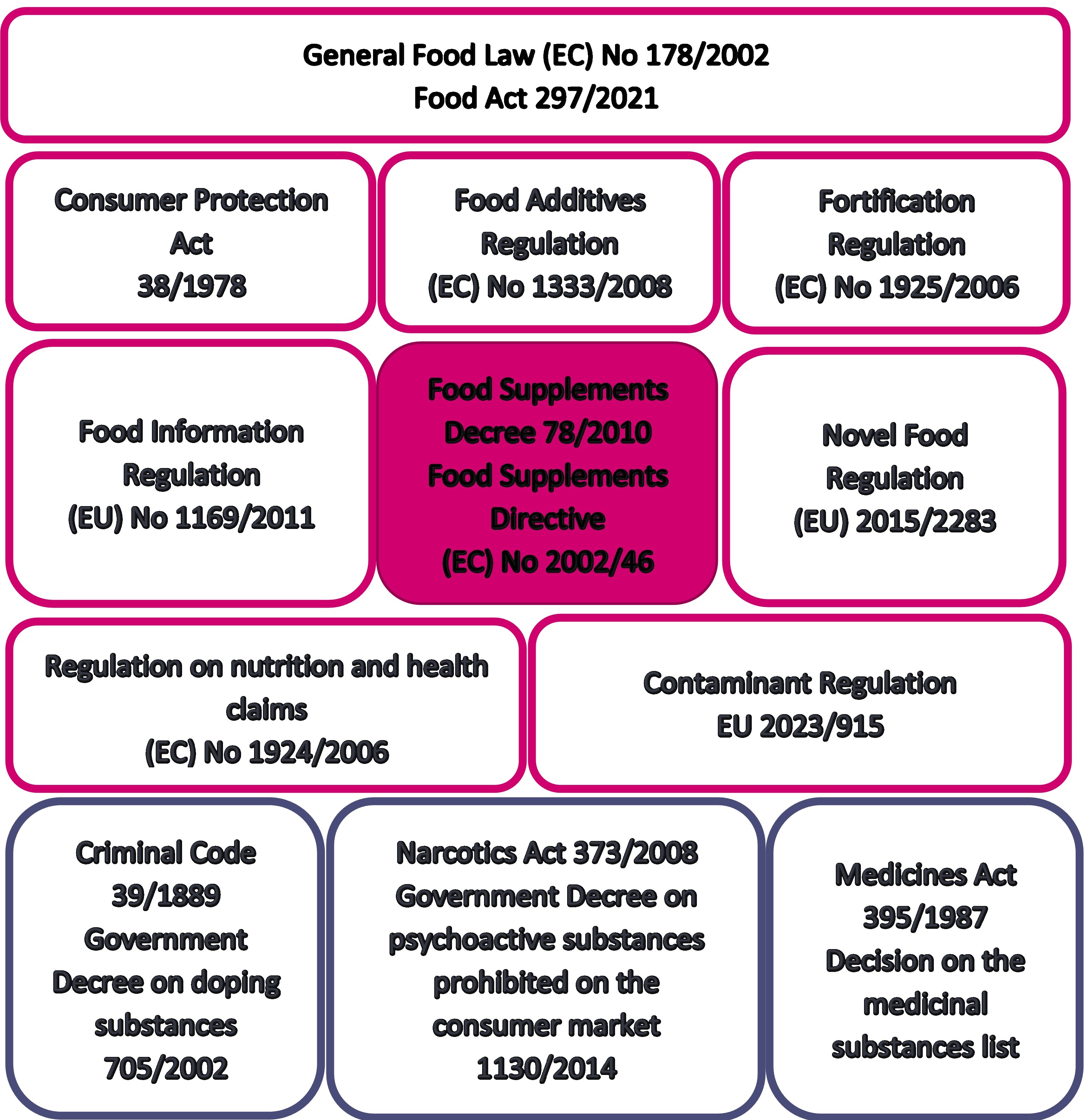
Figure 1. Key legislation on food supplements.
2 Definition of food supplement
In accordance with section 2 of Decree 78/2010 of the Ministry of Agriculture and Forestry on food supplements (in Finnish and Swedish only) (hereinafter Food Supplements Decree), food supplement is a pre-packaged product in the form of a briquette, capsule, pastille, tablet, pill, powder, concentrate, extract or liquid, or in some other equivalent dose form, that is marketed as a foodstuff. Food supplements are consumed in small unit quantities and the amount of energy they provide has no impact on the diet as a whole.
The Finnish Food Authority holds that the energy provided by a food supplement when consumed according to its recommended maximum dosage is not significant in terms of energy intake, provided that it does not exceed 200 kJ (50 kcal) per day. A small dose is one teaspoon or tablespoon (5-15 ml) of the product, which means that the maximum volume of a daily dose of the marketed product is 100 ml.
In addition, according to Article 2 of Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002 on the approximation of the laws of the Member States relating to food supplements (hereinafter Food Supplements Directive), ‘food supplements’ are concentrated sources of nutrients or other substances with a nutritional or physiological effect. Nutrients include vitamins and minerals. Other substances with a nutritional or physiological effect are considered to include, for example, fatty acids, amino acids and fibres as well as various plants, chemical substances or lactic acid bacteria.
The purpose of food supplements is to supplement the normal diet. They are not designed to replace a balanced diet. Products classified as medicinal products pursuant to the Medicines Act (395/1987) are not food supplements.
3 Composition of food supplements
Food supplements are composed of characteristic substances and, if necessary, any other ingredients, such as additives. The following paragraphs describe the ingredients used in food supplements in more detail.
3.1 Vitamins and minerals
Only the vitamins and minerals listed in Annex 1 to the Food Supplements Directive may be used in food supplements in the forms listed in Annex 2. The Directive does not apply to natural sources of vitamins and minerals.
The Annexes to the Directive are updated as needed and it is possible to request the addition of new substances and compounds to them. Requests for adding new vitamin or mineral compounds may be made by means of two different procedures, depending on whether the compound is a novel food or not:
- Compounds which have significant history of consumption in foods prior to 15 May 1997 and which are therefore not considered novel foods shall require the submission of an application to the European Commission for authorisation as a permitted source. European Commission guidance on submitting an application (pdf).
- Compounds which do not have significant history of consumption in foods prior to 15 May 1997 and are therefore considered novel foods shall require the submission of an application to the European Commission electronic submission system for authorisation as a novel food. Instructions for applying for a novel food authorisation on the Finnish Food Authority website.
Further information can be found on the European Commission website.
3.1.1 Purity criteria
The Decree on Food Supplements does not specify purity criteria for the vitamin and mineral compounds used in food supplements. The purity criteria defined in other Community legislation are applied to vitamin and mineral compounds. For example, the purity criteria specified for L-ascorbic acid used as an additive also apply to L-ascorbic acid used to fortify foods or in food supplements. The compounds of vitamins and minerals authorised as novel foods have been subject to specific purity criteria and are listed in the Union list of authorised novel foods (EU) 2017/2470 (hereinafter Union list). If Community level purity criteria have not been defined, it is also possible to use generally acceptable purity criteria commonly recommended by international bodies.
If Commission Regulation (EU) No. 231/2012 laying down specifications for food additives sets identification and purity criteria for compounds used in food supplements, these specifications shall be applied.
- If the aforementioned Regulation does not include these specifications, the identification and purity requirements recommended by the Codex Alimentarius Commission shall be applied. They are based on the identification and purity criteria of JECFA (The Joint FAO/WHO Expert Committee on Food Additives).
- The food additives for which the Codex Alimentarius Commission has recommended identification and purity criteria can be found on the FAO (Food and Agriculture Organization of the United Nations) website under the title “List of Codex advisory specifications for food additives”.
The “INS No." indicated for each additive can be used to search for purity criteria electronically from the “Combined Compendium of Food Additive Specifications” on the FAO website by entering the indicated “INS No." of the additive in the search field "INS Number". - If there are no identification and purity criteria specified in EU legislation or recommended by the Codex Alimentarius Commission, other criteria defined by JECFA shall be applied.
The identification and purity criteria recommended by Codex as well as other criteria defined by JECFA can be found electronically in the “Combined Compendium of Food Additive Specifications” as well as in publications FAO JECFA Monographs 1, Volume 1-3 (2005), FAO JECFA Monographs 3 (2006) and FAO JECFA Monographs 4 (2007), FAO, Rome. The determination methods used in identification and purity analyses can be found at the same URL address as well as in the publication FAO JECFA Monographs 1, Volume 4 (2005), FAO, Rome. - In the absence of the criteria referred to in Items 1-3, the purity criteria recommended by the European Pharmacopoeia shall be applied.
3.1.2 Minimum amounts
According to the Food Supplements Directive, minimum amounts shall be set appropriately to the daily dose recommended by the food supplement manufacturer. The minimum amounts of vitamins and minerals have not yet been laid down in legislation.
However, food supplements must be concentrated sources of nutrients, in which case minimum amounts may be applied as provided for in Regulation (EC) No. 1924/2006 of the European Parliament and of the Council on nutrition and health claims made on foods (hereinafter referred to as the Health and Nutrition Claims Regulation) to the claim “source of vitamins and/or minerals”.
Accordingly, a food supplement may be considered a source of a vitamin or mineral if its recommended daily dose contains a significant amount of that vitamin or mineral. The amount is considered significant, when the daily intake of the vitamin or mineral from the food supplement is 15 percent of the daily reference intake, when consumed according to the dosage instructions, regardless of whether the food supplement is in solid or liquid form. If the recommended daily dose of the food supplement is given as a range (e.g. 1-4 capsules), the Finnish Food Authority holds that the amount of the nutrient must also be significant in the lowest recommended daily dose (1 capsule).
In other words, in order for a food supplement to be marketed as a concentrated source of a vitamin or mineral, the food supplement, when consumed according to the dosage instructions, shall provide at least 15 percent of the daily reference intake of the vitamin or mineral concerned.
If the food supplement is sold specifically as an excellent or good source of nutrients, or marketed as being high in vitamins or nutrients (e.g. using the words "mega", "strong", "extra", "plus" in the name of the product), the daily intake from the food supplement, when consumed according to the dosage instructions, must be at least 30 percent of the daily reference intake.
The daily reference intakes (DRI/RI) of vitamins and minerals are listed in Table 1 and in Annex XIII of Regulation (EU) No 1169/2011 of the European Parliament and of the Council on the provision of food information to consumers (hereinafter Food Information Regulation).
3.1.3 Maximum amounts
According to the Food Supplements Directive, maximum amounts of vitamins and minerals present in food supplements per daily intake shall be set. So far, no maximum amounts have been set in legislation.
Food business operators shall verify on the basis of their own risk assessment that the level of vitamins or minerals in the food supplement which the operator produces or has produced, imports or distributes does not pose a health hazard. This is particularly emphasised in the case of nutrients for which the difference between the recommended intake and the upper limit of safe intake (safety margin) is narrow. Nutrients with a narrow safety margin are usually considered to include vitamins D and A, niacin, folic acid and minerals. With vitamin A, for example, tripling the recommended intake is enough to reach the upper limit of safe intake.
The European Food Safety Authority (EFSA) has assessed the safety of nutrients and set daily tolerable upper intake levels (UL) for certain vitamins and minerals. The UL indicates how much a given vitamin or mineral can be safely consumed in its entirety in continuous, long-term use (Table 1).
The UL takes into account the daily intake from all sources, including regular food, fortified food products, food supplements and certain vitamin and mineral preparations classified as medicines. An exception to this is magnesium, whose UL is based on only easily dissociable magnesium salts and MgO from food supplements, water and fortified food products. In other words, the UL set for magnesium does not include magnesium naturally present in foods and beverages.
When assessing the safety risk caused by a food supplement to the consumers, the potential targeting of a given food supplement to vulnerable consumer groups, such as children, pregnant and breastfeeding women and the elderly, shall be taken into account in addition to the UL.
The Finnish Food Authority holds that, if the recommended daily dose of the food supplement contains a vitamin or a mineral in an amount that exceeds the UL, the food supplement can be seen as posing a health hazard to consumers. If the recommended daily dose is given as a range (e.g. 1-4 capsules per day), exceeding the UL should be considered for the highest recommended daily dose (e.g. 4 capsules).
In such cases the operator who produces, imports or distributes the food supplement or has it produced, shall take measures to control the health hazard. The measures to be taken shall be determined on a case-by-case basis specific to the given conditions. Possible measures for controlling the hazard include, for example, reducing the amount of a given nutrient in the product, decreasing the daily dose, issuing a warning on the label or taking some other measure to ensure the safety of the food supplement for the consumer.
If such a food supplement is targeted at vulnerable consumer groups, the Finnish Food Authority will consider the food supplement in question a serious health hazard, which must result in the withdrawal of the product from the market.
More information on the withdrawal procedure can be found in Section 7.1.1 and on the Finnish Food Authority website.
For some nutrients, it has not been possible to establish an exact tolerable upper intake level (UL) because there is not enough information on them. Instead, the EFSA has provided an assessment of the safe level of intake for a given nutrient. This refers to the amount of a nutrient for which there is reasonably reliable evidence that it will not cause adverse effects.
Safe intake level is not as accurate as the UL value. If the safe intake level is exceeded, this does not automatically mean that it would cause harm. In addition, these values cannot be used to assess how many people would be at risk of suffering any adverse effects.
Table 1 summarises reference values for the daily intake of vitamins and minerals, minimum amounts, tolerable upper intake levels (UL). Table 2 shows safe levels of intake.
Table 1. Reference daily intake (RDI) 1 minimum amounts and tolerable upper intake levels 2,3,4,5,6,7,11 (UL).
| Nutrient (unit) | RDI |
Minimum amount |
UL (1-3) |
UL (4-6) |
UL (7-10) |
UL (11-14) |
UL (15-17) |
UL (18-) |
|---|---|---|---|---|---|---|---|---|
| Vitamins | ||||||||
| Vitamin A (µg) | 800 | 120 | 800 | 1100 | 1500 | 2000 | 2600 | 3000 |
| Vitamin D (µg) | 5 | 0,75 | 508 | 50 | 50 | 100 | 100 | 100 |
| Vitamin E (mg) | 12 | 1,8 | 100 | 120 | 160 | 220 | 260 | 300 |
| Vitamin K (µg) | 75 | 11,25 | - | - | - | - | - | - |
| Vitamin C (mg) | 80 | 12 | - | - | - | - | - | - |
| Thiamine (B1) (mg) | 1,1 | 0,165 | - | - | - | - | - | - |
| Riboflavin (B2) (mg) | 1,4 | 0,21 | - | - | - | - | - | - |
Niacin (B3) (mg)
|
16 | 2,4 |
2 |
3 220 |
4 350 |
6 500 |
8 700 |
10 900 |
| Vitamin B6 (mg) | 1,4 | 0,21 | 3,29 | 4,5 | 6,1 | 8,6 | 10,7 | 12,5 |
| Folic acid (B9) (µg) | 200 | 30 | 200 | 300 | 400 | 600 | 800 | 1000 |
| Vitamin B12 (µg) | 2,5 | 0,375 | - | - | - | - | - | - |
| Biotin (B7) (mg) | 50 | 7,5 | - | - | - | - | - | - |
| Pantothenic acid (B5) (mg) | 6 | 0,9 | - | - | - | - | - | - |
| Minerals | ||||||||
| Potassium (mg) | 2000 | 300 | - | - | - | - | - | - |
| Chloride (mg) | 800 | 120 | - | - | - | - | - | - |
| Calcium (mg) | 800 | 120 | - | - | - | - | - | 2500 |
| Phosphorus (mg) | 700 | 105 | - | - | - | - | - | - |
| Magnesium (mg) | 375 | 56,25 | - | - | - | - | - | 25010 |
| Iron (mg) | 14 | 2,1 | - | - | - | - | - | - |
| Zinc (mg) | 10 | 1,5 | 7 | 10 | 13 | 18 | 22 | 25 |
| Copper (mg) | 1 | 0,15 | 1 | 2 | 3 | 4 | 4 | 5 |
| Manganese (mg) | 2 | 0,3 | - | - | - | - | - | - |
| Fluoride (mg) | 3,5 | 0,525 | 1,611 | 2,0 | 2,0 | - | - | - |
| Selenium (µg) | 55 | 8,25 | 7012 | 95 | 130 | 180 | 230 | 255 |
| Chromium (µg) | 40 | 6 | - | - | - | - | - | - |
| Molybdenum (µg) | 50 | 7,5 | 100 | 200 | 250 | 400 | 500 | 600 |
| Iodine (µg) | 150 | 22,5 | 200 | 250 | 300 | 450 | 500 | 600 |
1 Food Information Regulation (EU) No. 1169/2011 Annex XIII
2 Tolerable Upper Intake Levels for Vitamins and Minerals, Scientific Committee on Food, Scientific Panel on Dietetic Products, Nutrition and Allergies, EFSA, February 2006.
3 Scientific Opinion on the Tolerable Upper Intake Level of vitamin D, EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA), EFSA Journal 2012; 10 (7):2813.
4 Scientific Opinion on the update of Tolerable Upper Intake Level for vitamin D for infants, EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA), EFSA Journal 2018; 16 (8): 5365.
5 Scientific opinion on the tolerable upper intake level for selenium, EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA), EFSA Journal 2023;21(1):7704.
6 Scientific opinion on the tolerable upper intake level for vitamin B6, EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA), EFSA Journal 2023;21(5):8006.
7 Scientific opinion on the tolerable upper intake level for vitamin D, including the derivation of a conversion factor for calcidiol monohydrate, EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA), EFSA Journal 2023;21(8):8145
8 The tolerable upper intake level for vitamin D is 25 μg for infants 0-6 months of age and 35 μg for infants 6-12 months of age.
9 The tolerable upper intake level for vitamin B6 is 2.2 mg for infants 4-6 months of age and 2.5 mg for infants 7-11 months of age.
10 Only magnesium salts and MgO contained in food supplements, fortified food products and water are taken into account.
11 Updated consumer risk assessment of fluoride in food and drinking water including the contribution from other sources of oral exposure, EFSA Scientific Committee, EFSA Journal. 2025;23:e9478. The tolerable upper intake level for fluoride applies to the following age groups: 1 mg for those under 1 year of age, 1.6 mg for those between 1 and 3 years of age and 2 mg for those between 4 and 8 years of age.
12 The tolerable upper intake level for selenium is 45 µg for infants 4-6 months of age and 55 µg for infants 7-11 months of age.
Table 2. Safe intake levels for some nutrients 1, 2, 3.
| Nutrient (unit) | 4-11 months |
1-3 years old |
4-6 years old | 7-10 years old | 11-14 years old | 15-17 years old | Adults |
|---|---|---|---|---|---|---|---|
| Iron (mg) | 5 | 10 | 15 | 20 | 30 | 35 | 40 |
| 4-11 months | 1-2 years old | 3-6 years old | 7-13 years old | 14-17 years old | Adults | ||
| Manganese (mg) | 2 | 4 | 5 | 6 | 7 | 8 | |
| 4-11 months | 1-3 years old | 4-8 years old | 9-17 years old | Adults | |||
| Fluoride (mg) | - | - | - | 3,3 | 3,3 | ||
1 Scientific opinion on the tolerable upper intake level for iron, EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA), EFSA Journal. 2024;22:e8819.
2 Scientific opinion on the tolerable upper intake level for manganese, EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA), EFSA Journal. 2023;21:e8413.
3 Updated consumer risk assessment of fluoride in food and drinking water including the contribution from other sources of oral exposure, EFSA Scientific Committee, EFSA Journal. 2025;23:e9478. The safe intake level of fluoride for children over 8 years of age and adults is 3.3 mg.
3.2 Other substances with nutritional or physiological effects
In addition to nutrients, other substances used in food supplements include, for example, fatty acids, amino acids, plants, plant extracts, herbs, bee products, enzymes and lactic acid bacteria. No provisions regarding other substances or their concentrations have yet been laid down in legislation pertaining to food supplements However, food supplements are subject to other existing legislation, such as Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods (hereinafter the Novel Food Regulation) and Regulation (EC) No 1925/2006 of the European Parliament and of the Council on the addition of vitamins and minerals and of certain other substances to foods (hereinafter the Fortification Regulation).
According to the Novel Food Regulation, foods that had not been consumed to a significant degree by humans in the EU before 15 May 1997 may not be placed on the market until they have been approved and included on the Union list. Consequently, ingredients which had not been consumed to a significant degree by humans in the EU before 15 May 1997 shall not be used in the manufacture of food supplements until they have been approved and included on the Union list. For more information on novel foods, see Section 3.5.
The Fortified Foods Regulation provides for a procedure to restrict or prohibit the use of certain substances in food. A procedure provided for in Article 8 of the Fortified Foods Regulation can be followed to effect a complete prohibition or restriction of the use of a substance that poses a potential health risk. Substances prohibited and restricted under the Fortified Foods Regulation are described in section 3.12.
Although as a rule there are no laws in Finland that would restrict the use of other substances in food supplements, it should be kept in mind that other EU member states may have lists of permitted and prohibited plants or other substances in place. These are permitted until there Community-level legislation is enacted on the matter.
However, the general requirements specified for foodstuffs also apply to food supplements and their ingredients:
- they shall be safe in terms of their microbiological, chemical and physical quality.
- they shall have a significant history of consumption as a conventional food or food supplement in the EU before 15 May 1997 or alternatively an appropriate novel food authorisation.
- they may not turn the product into a medicine.
3.3 Raw materials and products of animal origin
Raw materials of animal origin, such as fish oil or colostrum, may be used in some food supplements if they meet the requirements laid down in other legislation, such as the Novel Food Regulation. Raw materials of animal origin fall within the scope of the Food Act (297/2021), Regulation (EC) No. 853/2004 of the European Parliament and of the Council laying down specific hygiene rules for food of animal origin, and the more specific provisions issued under them. The control of the conditions of the production of raw materials containing the raw materials in question is based on these. More detailed import requirements are also applied to raw materials and foodstuffs of animal origin, which are described in more detail below.
3.3.1 Imports from internal market
When raw materials or foodstuffs of animal origin are imported to Finland from EU countries in the internal market, this is considered a food business, which must be registered to the food control authority of one’s own municipality. The intra-EU imports of foodstuffs of animal origin are controlled as part of the Oiva food control inspections conducted by municipalities.
More information on the intra-EU import of food of animal origin can be found on the Finnish Food Authority website.
3.3.2 Imports from third countries
Food supplements containing ingredients of animal origin are usually classified as so-called composite products. According to Commission Delegated Regulation (EU) 2022/2292, composite products are foodstuffs containing both products of plant origin and processed products of animal origin. In this case, food supplements must meet the requirements set for composite products.
Food supplements that are imported from countries outside the EU and that are packaged for the end consumer and contain processed products of animal origin (including glucosamine, chondroitin or chitosan) are exempt from official controls at border control posts (i.e. veterinary border inspections), provided they meet all the requirements listed in Article 3 of Regulation (EU) 2021/630.
If a composite product contains gelatine or collagen from ruminant bones, an EU Health Certificate for composite products is required (Chapter 50: Model "COMP" (EU) 2020/2235). In this case, private attestation provided by a so-called operator is not sufficient (private attestation, i.e. Annex V (EU) 2020/2235).
More detailed information on the requirements for composite products can be found on the Finnish Food Authority website.
If food supplements imported from outside the EU contain only products of animal origin, they must meet the import requirements for the ingredient in question. Examples of such products
- milk products (in Finnish) (powdered milk, whey, lactose or other milk ingredients)
- egg products (in Finnish) (e.g. powdered eggs)
- meat products (in Finnish) (all animal tissue, such as thymus and other glands or ground bone material, including extracts)
- fishery products (in Finnish) (such as shell or bone meal, fish oil).
If gelatine capsules (hard or soft) imported from outside the EU are made from ruminant bones, a separate EU Health Certificate is required (Chapter 41: Model "GEL" (EU) 2020/2235). In other words, if the gelatine capsule of the product is made from ruminant bones and the capsules contain fish oil, two separate EU Health Certificates are required (Chapter 41: Model "GEL" and Chapter 28: Model "FISH-CRUST-HC").
More information on third country imports is available on the Finnish Food Authority website (in Finnish).
3.3.3 Seal products
Trade in seal products, such as seal oil, is governed by Regulation (EC) No. 1007/2009 on trade in seal products, and Regulation (EU) No. 737/2010 laying down rules for its implementation. The placing of seal products on the market is only allowed provided the conditions set out in Regulation No. 1007/2009/EC are fulfilled and the seal product is accompanied by the certificate referred to in Regulation No. 737/2010/EU.
Trade in seal products is, in addition to EU Regulations, also restricted by a national act (1107/1996), which forbids the importation of raw skins and processed skins of whitecoat new-born harp seals and newborn hooded seals, as well as the importation of products manufactured from these skins. In this context, importation refers to all products brought into Finland, regardless of the manner in which they are brought, including from another EU member state.
Trade in seals is in Finland controlled by the police and the Customs.
3.4. Food improvement agents
Food improvement agents refer to additives, flavourings and enzymes used in foods. Additives and enzymes are always used for some technological purpose. Flavourings are used to improve or change the aroma and/or flavour of the food.
Regulation (EC) No 1333/2008 on food additives lists the additives approved for use in the production of food supplements and the maximum amounts of these additives in Annex II, under food group 17 (excluding food supplements intended for infants and small children - no additives whatsoever may be used in the production of these food supplements). Food supplements are in group 17 divided into solid and liquid food supplements based on their form. The approved additives and their maximum amounts are specified for each form of food supplements. Annex III to the Food Additives Regulation lists the additives and carriers that are permitted for use in additives, enzymes, flavourings and nutrients as well as the conditions for their use.
The additives and carriers used shall comply with the specifications laid down in Commission Regulation (EU) No. 231/2012 concerning origin, purity criteria and other required information.
The flavourings that are permitted for use in foods (incl. food supplements) are listed in Regulation (EC) No. 1334/2008 of the European Parliament and of the Council on flavourings and certain food ingredients with flavouring properties for use in and on foods (hereinafter Flavouring Regulation). The enzymes that are permitted for use in foods will be listed in Regulation (EC) No. 1332/2008 of the European Parliament and of the Council on food enzymes.
The Finnish Food Authority has prepared a guideline for the control of food improvement agents for food authorities and food business operators. The guideline can be found on the Finnish Food Authority website (in Finnish and Swedish only).
More information on food improvement agents can be found on the Finnish Food Authority website (in Finnish and Swedish only).
3.5 Novel foods
According to the Novel Food Regulation (EU) 2015/2283, novel foods are those not used for human consumption to a significant degree within the European Community prior to the adoption of the Regulation on 15 May 1997. Such foods or their ingredients may include, for example, wild plants with no commonly known use in foods, exotic plants from non-EU countries, new extracts derived from a food of animal origin or a plant as well as new synthetic food ingredients.
Novel foods may not be placed on the market within the EU without a safety assessment and novel food authorisation from the European Commission. An authorisation is also required for new food and ingredient production processes (such as foods produced using nanotechnology), if the production process was not in use prior to 15 May 1997.
The Regulation on novel foods also applies to food supplements and their ingredients. Consequently, the authorisation referred to in the Novel Food Regulation is required for any food supplements containing ingredients that have not been used as normal foods or food supplements within the Community prior to 15 May 1997. If, substances and plants have a substantiated history of use only as food supplements prior to 1997, they can still be used in food supplements, but novel food authorisation will be required if use of the substance or plant in question is expanded to other food groups.
Food business operators are obligated to clarify and, if necessary, provide substantiation on the history of consumption of their products and ingredients as a food prior to 1997. If the history of consumption cannot be substantiated, the products and their ingredients shall be basically considered novel foods and thus fall within the scope of the Novel Food Regulation.
There is no comprehensive list of ingredients considered to be novel foods. The Novel Food Status Catalogue maintained by the European Commission contains information on the novel food status of various foods. The Novel Food Status Catalogue is only indicative and the absence of a food from the list does not mean that it has a history of use as food and is consequently not a novel food, or vice versa. Also, the classification of a product may change if new and reliable information emerges regarding its history of use. The Finnish Food Authority website also contains other public sources of data that can be used to establish the history of use of a food and its status as a novel food.
Authorised novel foods are listed on the Union list, together with their conditions of use and specifications. The use of these novel foods is permitted in foods in the manner specified in the conditions of use. The Union list can be found on the European Commission website.
Additional information on novel foods can be found on the Finnish Food Authority website.
3.6 Genetically modified ingredients
According to Regulation (EC) No. 1829/2003 of the European Parliament and of the Council on genetically modified food and feed, neither genetically modified organisms (GMOs) nor foods may be cultivated or placed on the market in the EU unless they are authorised. Some genetically modified plants (maize, soya bean, rapeseed, cotton, sugar beet) have been authorised on the EU market since 1996. Only one genetically modified maize variety has been authorised for cultivation in the EU. The Regulation also applies to food supplements.
Authorised genetically modified products and ingredients placed on the market must be traceable in accordance with Regulation (EC) No 1830/2003 of the European Parliament and of the Council concerning the traceability and labelling of genetically modified organisms and the traceability of food and feed products produced from genetically modified organisms. In every stage of placing genetically modified products on the market, the receiving operator shall be informed in writing of the genetically modified nature of the product and the unique identifier assigned to the genetically modified variety in question. Information on genetically modified varieties authorised in the EU and their unique identifiers can be found in the Community register of genetically modified food and feed. Additional information on the authorisation of genetically modified products (in Finnish and Swedish only) and control guidelines for genetically modified food can be found on the Finnish Food Authority website.
Genetically modified ingredients must be indicated on the labelling of food supplements. The label of pre-packaged products consisting of or containing GMOs shall be marked “Product contains GMOs” or “Product contains genetically modified organisms [name of one or more of the organisms]”. Labelling requirements are laid down in Regulation (EC) No. 1830/2003.
In the own-check and control of food supplements, consideration should also be given to so-called high-risk ingredients with regard to genetically modified ingredients, i.e. genetically modified varieties present on markets outside the EU but not approved in the EU. These high-risk ingredients include, in particular, soya beans and maize grown in the United States, rapeseed grown in Canada, rice grown in China and papaya grown in Thailand or the United States. In performing its own-check, an operator must ensure, either by means of documents or analyses, that high-risk ingredients used in food supplements do not contain genetically modified varieties that are not approved in the EU. For genetically modified varieties authorised in the EU, steps must be taken to ensure that the varieties used in the products correspond to the description (whether they are genetically modified or not) and that any genetically modified ingredients are indicated on the labelling in accordance with Regulation (EC) No. 1829/2003.
The requirements also apply to varieties produced using new genome-editing techniques at the time of publication, whether or not they can be produced using traditional plant breeding techniques. The Commission’s proposal for a regulation on plants produced using certain new genome-editing techniques is still being drafted as the guidelines are being updated.
3.7 Medicinal and herbal medicinal products
Under section 6 of the Medicines Act (395/1987), the Finnish Medicines Agency Fimea (hereinafter Fimea) will decide whether a substance or product is considered a medicinal product. Each product is specifically classified based on an assessment of its influence and intended use, as referred to in section 3 of the Medicines Act.
Under section 83 of the Medicines Act, Fimea has compiled a medicinal products list, which is an indicative list of the substances and herbal medicinal products that are in medicinal use in Finland (Annexes 1 and 2). The medicinal products list is not exhaustive - substances and herbal medicinal products not included on the list can also be considered medicines, provided that they meet the definition of a medicinal product under the Medicines Act.
The salts and esters of the medicinal substances included in the list are not listed, but they are biologically comparable to medicinal substances. The medicinal products list also includes Annex 1A, which is a list of medicinal substance analogues and prohormones. Because these substances are always considered on the basis of their influence to be comparable to prescription-only medicines, any food supplements containing these substances will be classified as medicines.
Classification as a medicine is always based on the assessment of a specific product. In addition to the medicinal products list, norms issued at the EU level shall also be taken into account in the classification procedure. At Fimea, the classification of herbal medicinal products containing or produced from herbal medicinal products and (traditional) herbal medicinal products is guided by, for example, European Union (EU) herbal monographs and EU list entries. Fimea recommends that food business operators examine the information published on the websites of the European Commission and the European Medicines Agency, if their products contain substances of plant origin, i.e. herbal medicinal products or herbal medicinal products made from them (e.g. extracts, tinctures, pressed juices) as a characteristic substance.
Scientific evaluations of the European Medicines Agency (EMA) Committee on Herbal Medicinal Products (HMPC) and European Union plant monographs and public statements are used to classify herbal medicinal products and traditional herbal medicinal products by the national authorities of EU Member States.
In addition, the European Union Community Catalogue is valid throughout the EU and guides the classification of traditional herbal medicinal products at Fimea for substances of plant origin, herbal medicinal products and combinations thereof.
Additional information on the matter can be found on the European Medicines Agency website and European Commission website.
Food regulations do not restrict the use of substances or herbal medicinal products included on the medicinal products list as ingredients of food supplements, provided that their use is not based on their medicinal properties. In other words, there may be substances on the market that are identical or products that are made from the same plants, but different requirements are applied to them depending on whether they are placed on the market in accordance with regulations on medicinal products or food regulations.
The Finnish Food Authority recommends contacting Fimea to assess the need for classification if a food supplement contains substances on the medicinal products list or if medicinal products listed on the medicinal products list have been used in the manufacture of the food supplement. Fimea may classify a product as a medicine also at its own initiative if the product meets the criteria defined for a medicine.
The medicinal products list and additional information on classification can be found on the Fimea website.
3.8 Alcohol
Because alcohol is used as an extraction solvent in, for example, the production of herbal preparations, food supplements may contain significant amounts of alcohol. According to Food Information Regulation (EU) No. 1169/2011, the alcohol content of liquid foods must be declared, if it exceeds 1.2 percent by volume. The Finnish Food Authority recommends that the alcohol content of solid foods also be declared if it is higher than 1.8 percent by weight.
According to the Finnish Alcohol Act (1143/1994), an alcoholic substance is a substance or product which contains at least 2.8 percent of ethyl alcohol by volume. An alcoholic beverage is an alcoholic substance intended for drinking that contains a maximum of 80 percent ethyl alcohol by volume and an alcoholic preparation is an alcoholic substance that is not an alcoholic beverage.
Food supplements classified as alcoholic preparations may only be produced by companies that have been granted a production licence for alcoholic beverages and spirits by the Finnish Supervisory Agency. Alcohol preparations may only be imported for commercial purposes under a wholesale licence granted by the Agency. Retail outlets do not need a licence to sell alcoholic food supplements.
The sale and marketing of alcoholic food supplements is free under the Alcohol Act, but their manufacturer, importer or seller must not promote their use for the purpose of intoxication. The Agency may prohibit the release of an alcoholic substance on the market or impose an obligation to remove an alcoholic substance from the market without compensation if the product or its presentation violates the provisions and regulations issued for it. In other words, although no licence is required for the retail sale of alcohol, it can become subject to control measures.
Solid foods containing up to 2.8 percent alcohol by weight, such as chocolate confectionery or ice cream, may be sold freely. The provisions of the Alcohol Act on alcoholic beverages, such as age limits and licence requirements, apply to the retail sale and marketing of stronger products. The control of these products in accordance with food legislation is the responsibility of municipal food control authorities.
More information on alcoholic products can be found on the Agency’s website.
3.9 Natural toxins
Some plants may naturally contain various harmful compounds, or toxins, which the plant uses as protection against harmful insects and diseases or against perishing. When used in food, the natural toxins of plants may have adverse effects on health. The type of toxin, the level of the toxin in the edible part of the plant and the individual sensitivity of people to different substances determine whether the toxin causes any symptoms. In some cases, the level of harmful substances can be influenced through appropriate processing. Some toxins, such as gyromitrin in false morels, are water soluble or volatile and will therefore break down when boiled.
Commission Regulation (EU) 2023/915 on maximum levels for certain contaminants in food (hereinafter Contaminant Regulation) sets maximum levels only for some natural toxins. However, food business operators are responsible for the safety of the foods they sell and market, and for the safety of the ingredients of the foods, also with respect to any natural toxins. Food business operators who produce or market foods in which e.g. plants containing natural toxins are used shall carry out a risk assessment process to verify the safety of the food.
The European Food Safety Authority EFSA has compiled a list (EFSA Compendium of Botanicals) of plants known to contain naturally occurring substances of concern that are e.g. toxic, addictive or psychotropic. The purpose of the Compendium is to support risk assessment on food supplements in regard to their ingredients of plant origin, by identifying the naturally occurring compounds of concern which should be the focus of the assessment. The Compendium does not state whether the compounds of concern can be removed from the plant by means of, for example, processing.
Food supplements are concentrated sources of their characteristic ingredients (nutrients or other substances with a nutritional of physiological effect). As a result of this, any harmful compounds contained in a food supplement are also in a concentrated form. The potential concentration of natural toxins in the food supplement shall thus be taken into account in a risk assessment.
Additional information on natural toxins can be found on the Finnish Food Authority website.
3.10 Contaminants
Contaminants in foodstuffs refer to substances that have not been intentionally added to foods, but may nevertheless occur as a result of, for example, production or other processing stages, or due to environmental contamination. Contaminants include mycotoxins (mould toxins), heavy metals, dioxins and PCBs and PAHs. In sufficiently high concentrations, contaminants can render food harmful to human health or make it unfit for consumption. Maximum levels (ML) have been set out for some contaminants occurring in foods, and food business operators shall observe these levels when performing their own-check. Authorities perform risk-based control on the implementation of the operators’ own-check.
High levels of lead, cadmium and mercury have been found in certain food supplements, such as plant and herbal preparations. These food supplements may significantly increase exposure to, for example, heavy metals. It is for this reason that maximum levels have been set for the levels of lead, cadmium and mercury in food supplements. The maximum levels are safe and as low as reasonably achievable by following good manufacturing practices. In particular, food supplements that consist completely or primarily of dried seaweed or mussels (e.g. greed-lipped mussel) may contain high levels of cadmium, as it is naturally accumulated in seaweed and mussels. The maximum level set for cadmium is therefore higher.
When food or a food ingredient spoils, mycotoxins may be produced. A mycotoxin called citrinin may occur in rice fermented withred yeast (Monascus purpureus), for example. Because of this, some red yeast rice preparations may contain high levels of citrinin, which can be harmful to e.g. the kidneys. A maximum level has therefore been set also for the citrinin content of red yeast rice preparations.
Regulations pertaining to food contaminants (Contaminants Regulation (EU) 2023/915 and amendments) are under constant development, also with respect to food supplements. Consequently, food business operators need to monitor changes in regulations and take into account the valid maximum levels of different contaminants when performing their own-check.
Raw materials exceeding the maximum levels laid down in the legislation may not be used as ingredients in foodstuffs, such as food supplements.
Table 3. Examples of possible contaminants in different types of food supplements, and maximum levels of the contaminants (applicable to marketed food supplements)
| Contaminant | Maximum amount | Type of food supplement | ||
|---|---|---|---|---|
| Dioxins and PCBs | Dioxins total | Dioxins and dioxin-like PBC compounds, total | Non-dioxin-like PCB compounds | |
| 1,75 pg/g fat | 6,0 pg/g fat | 200 ng/g fat | Food supplements containing marine oil | |
| Lead | 3,0 mg/kg | All food supplements | ||
| Cadmium | 1,0 mg/kg | All food supplements, excluding those consisting of at least 80 percent dried seaweed, products derived from seaweed or dried mussels | ||
| 3,0 mg/kg | Food supplements consisting of at least 80 percent dried seaweed, products derived from seaweed or dried mussels | |||
| Mercury | 0,10 mg/kg | All food supplement | ||
| Citrinin | 100 µg/kg | Food supplements based on rice fermented with red yeast (Monascus purpureus) | ||
| Pyrrolizidine alkaloids | 400 μg/kg | Food supplements containing a preparation of plant origin, including extracts, excluding pollen-based food supplements | ||
| 500 μg/kg | Pollen-based food supplements | |||
| PAHs | Benzo(a)pyrene | Sum of benzo(a)pyrene, benzo(a)anthracene, benzo(b)fluoranthene and chrysene | ||
| 10,0 µg/kg | 50,0 µg/kg | Food supplements containing substances of plant origin and preparations derived from them, and food supplements containing propolis, royal jelly or spirulina algae, or preparations derived from them | ||
| 2,0 µg/kg | 10,0 µg/kg | Vegetable oils used as a food supplement | ||
3.11 Pesticide residues
Plant protection products, or pesticides, refer to products used in plant production to e.g. ward off weeds, protect crops from insects and other pests, prevent plant diseases, regulate plant growth or improve the durability of the products after harvest. When pesticides are used in plant production, it is possible that residues of these products will be found in foods. Maximum pesticide residue levels have been set for various foodstuffs (foods of both plant and animal origin) and food business operators must observe these in their in-house control. Authorities supervise the own-check performed by operators through risk-based random testing.
Raw materials exceeding the maximum levels laid down in the legislation may not be used as ingredients in foodstuffs, such as food supplements. Analyses conducted by authorities have found pesticide residues in e.g. food supplements that contain oils or herbs.
Regulation (EC) No 396/2005 of the European Parliament and of the Council on maximum residue levels of pesticides in or on food and feed of plant and animal origin (incl. Annexes) specifies the maximum pesticide residue levels in foods of plant and animal origin. The current maximum residue limits for each product and active substance described in the Annexes to the Regulation are easiest to find in the EU pesticide database.
3.12 Prohibited and restricted substances
Although Finnish legislation has no specific list of substances that may not be used in food supplements, the use of some substances is generally prohibited in food based on other regulations.
3.12.1 Substances referred to in Article 8 of Regulation (EC) No 1925/2006 (Fortification Regulation)
Article 8 of Regulation (EC) No 1925/2006 provides for a procedure that may be used to impose an EU-wide prohibition or restriction on the addition of certain other substances to food if they represent a potential health risk. An up-to-date list of prohibited and restricted substances can be found on the Finnish Food Authority website. Below are examples of substances used in food supplements whose use is prohibited or restricted:
Under the current regulatory procedure, the use of the following substances in foodstuffs is currently prohibited:
- The herb Ephedra and preparations derived from it which are of the genus Ephedra
- Yohimbe bark and its preparations originating from Yohimbe (Pausinystalia yohimbe (K. Schum) Pierre ex Beille)
- Aloe-emodin and all preparations in which this substance is present
- Danthron and all preparations in which this substance is present
- Emodin and all preparations in which this substance is present
- Preparations from the leaf of Aloe species containing hydroxyanthracene derivatives.
The use of the following substances as food is restricted:
- Green tea extracts containing (-)- epigallocatechin-3-gallate (excluding aqueous green tea extracts containing (-)- epigallocatechin-3-gallate which after reconstitution in beverages have a composition comparable to traditional green tea infusions).
- Daily portion of food shall contain less than 800 mg of (-)-epigallocatechin-3-gallate.
- The label shall
- provide the maximum number of portions of the food for daily consumption and a warning not to consume a daily amount of 800 mg of (-)-epigallocatechin-3-gallate or more.
- The label shall indicate the content of (-)- epigallocatechin-3-gallate per portion of the food.
- The label shall include the following warnings:
- “Should not be consumed if you are consuming other products containing green tea on the same day”
- “Should not be consumed by pregnant or lactating women and children below 18 years old”
- “Should not be consumed on an empty stomach”
- Monacolins from red yeast rice
- Individual portion of the product for daily consumption shall provide less than 3 mg of monacolins from red yeast rice.
- The label shall
- provide the number of individual portions of the product for maximum daily consumption and a warning not to consume a daily amount of 3 mg of monacolins from red yeast rice or more.
- The label shall indicate the content of monacolins per portion of the product.
- The label shall include the following warnings:
- “Should not be consumed by pregnant or lactating women, children below 18 years old and adults above 70 years old”.
- “Seek advice from a doctor on consumption of this product if you experience any health problems”.
- “Should not be consumed if you are taking cholesterol-lowering medication”.
- “Should not be consumed if you are already consuming other products containing red yeast rice”.
Additional information can be found on the Finnish Food Authority website and the European Commission website.
3.12.2 Caffeine
In 2015, the European Food Safety Authority (EFSA) delivered its Scientific Opinion on the safety of caffeine. According to the Opinion, single doses of caffeine up to 200 mg (approximately 3 mg/kg body weight) and daily doses of caffeine up to 400 mg a day (5.7 mg/kg body weight) do not give rise to safety concerns for healthy adults in the general population (taking all sources of caffeine into account). Caffeine intakes from all sources up to 200 mg per day consumed throughout the day during pregnancy or breastfeeding do not give rise to safety concerns for the foetus or infant. There is insufficient data on the safe intake of caffeine in children and adolescents to specify safe intake limits. However, according to the EFSA Opinion, safe intake values derived from adult body weight in kilograms may be applied to children. Thus, the limit value for children (1-17 years) can be set at a maximum of 3 mg of caffeine per kilogram of body weight per day.
Based on the EFSA Opinion, the Finnish Food Authority holds that a single dose of a food supplement for adults (e.g. tablet) may contain up to 200 mg of caffeine, thus making the recommended daily dose of caffeine 400 mg.
The warning labels required for food supplements containing caffeine are described in section 4.4.
3.12.3 Hormones and doping substances
The use of hormones and other doping substances is prohibited in food supplements as it is in all other food products. Doping refers to the enhancement of athletic performance by methods alien to the human body, such as taking medicines. Doping may improve athletic performance, but it may also severely damage the athlete's health.
According to chapter 44(6) of the Finnish Criminal Code (39/1889), doping offences include the manufacture, import and distribution of doping substances. According to chapter 44(16) of the Criminal Code, the following are considered doping substances:
- synthetic anabolic steroids and their derivatives
- testosterone and its derivatives
- growth hormones; and
- chemical substances that increase the production of testosterone, its derivatives or growth hormone in the human body.
Government decree 705/2002 provides a more detailed listing of doping substances than in chapter 44(16) of the Criminal Code.
Each year, the Finnish Center for Integrity in Sports (FINCIS) publishes its “List of Prohibited Substances and Methods in Sports". The list provides a much more comprehensive classification of doping substances than found in the Criminal Code. The sports sector therefore has more stringent regulations than the Criminal Code.
3.12.4 Narcotic and psychotropic substances
According to Article 2 of Regulation (EC) No 178/2002 of the European Parliament and of the Council laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety, the definition of food does not include narcotic or psychotropic substances within the meaning of the United Nations Single Convention on Narcotic Drugs, 1961, and the United Nations Convention on Psychotropic Substances, 1971. Their use is thus strictly prohibited in food (incl. food supplements).
Substances used solely for purposes of intoxication, or designer drugs, are controlled under the Narcotics Act (373/2008). In the Narcotics Act, designer drugs are defined as psychotropic substances that are banned from the consumer market. According to the Narcotics Act, these substances are used for intoxication purposes, and they are potentially hazardous to the health. The aforementioned psychotropic substances are listed in Government Decree (1130/2014). The Decree prohibits the production, storage, sale and distribution of psychoactive substances banned from the consumer market as well as their import into Finland.
3.12.5 Endangered species in violation of CITES Convention
Endangered species in violation of the CITES Convention may not be used as ingredients for food supplements. Entering into force in 1975, CITES (Convention on International Trade in Endangered Species of Wild Fauna and Flora) regulates international trade in over 35,000 endangered plant and animal species (e.g. whales, certain species of sharks), parts thereof and products made from them. CITES has been ratified by more than 160 countries. In Finland, the Convention entered into force in 1976.
CITES has been implemented in EU member states through the EU’s own CITES legislation. In addition to cross-border trade, EU legislation also regulates intra-EU trade in endangered species. Trade refers to all commercial exploitation, i.e. purchasing, offering to purchase, acquiring for commercial purposes, displaying for commercial purposes, exploiting for gain and sales, possessing for the purpose of sale, offering for sale or transporting for the purpose of sale.
International cross-border movement of CITES species is mainly regulated by a range of import and export restrictions, depending on the degree to which a species is threatened and how much use its populations can endure. In practice, trade is controlled based on written permits that are issued by the environmental authorities of each country (in Finland, the Finnish Environment Institute) and that need to be presented to Customs.
Additional information on the CITES Convention, related permits and legislation.
4 Information to be provided on food supplements
4.1 General requirements
According to Article 7 of Regulation (EU) No. 1169/2011 of the European Parliament and of the Council on the provision of food information to consumers (Food Information Regulation):
- Food information shall not be misleading
- Food information shall be accurate, clear and easy to understand for the consumer.
- Subject to derogations provided for by Union law, food information shall not attribute to any food the property of preventing, treating or curing a human disease, nor refer to such properties.
The above requirements shall apply not only to the labelling of the food but also to the advertising and presentation of foods, in particular their shape, appearance or packaging, the packaging materials used, the way in which they are arranged and the setting in which they are displayed.
According to Article 8 of the Food Information Regulation, the food business operator responsible for the food information shall be the operator under whose name or business name the food is marketed or, if that operator is not established in the Union, the importer into the Union market.
For pre-packed foods, the mandatory particulars shall appear on the pre-packaging or on a label attached thereto. The food information shall be marked in such a way as to be easily visible, with sufficiently large print, clearly legible, understandable and indelible. As a rule, labelling on food supplements must be in Finnish and in Swedish (Decree 834/2014 of the Ministry of Agriculture and Forestry on the provision of food information to consumers). Exceptions to this include food supplements sold locally in a monolingual municipality, where labelling shall be at least in the language of the municipality.
4.1.1 Information to be provided on food supplements in distance selling
Distance selling refers to marketing using a means of distance communication. These means of distance communication can refer to any means that can be used to conclude a contract between a business and consumer without having these two parties be present at the same location at the same time (Article 2 of the Food Information Regulation). Distance selling covers e.g. sales by internet and telephone, mail order sales or sales on internet sales platforms, in social media or teleshopping.
Article 14 of the Food Information Regulation lays down the mandatory information to be provided on prepacked foods in distance selling. Almost the same mandatory food information shall be available to consumer on foods offered for sale by means of distance communication as on foods marketed through normal channels, with the exception of the minimum durability date and the "use by" date. The information shall be provided without the food business operator charging consumers supplementary costs. All mandatory particulars shall be available at the moment of delivery. The mandatory information shall be provided in both Finnish and Swedish on food supplements sold through distance selling. For example, an image of the packaging label may be a sufficient way to present mandatory information, provided that the information is clear and legible.
4.2 Mandatory food information
According to Article 9 of the Food Information Regulation, the following information is to be provided for foods (including food supplements):
- Name of the product
- The name provides a brief and accurate description of the food product contained in the packaging (e.g. calcium tablet, oat shoot extract). The term "food supplement", which must also be shown, is not in itself sufficient as a name. A trademark or trade name cannot be used to replace the name of a food product. The trade name or part of the name of a food supplement must not be the same as the name of a medicinal product. A name similar to a medicine can also give the impression that a food supplement has medicinal properties.
- Quantity of content
- The content shall be expressed in units of volume for liquid products and units of weight for other foods.
- In addition, the Finnish Food Authority recommends that the number of food supplements in solid form (tablets, capsules, etc.) be indicated on the packaging.
- Name or business name and address of the operator responsible for food information
- List of ingredients
- An ingredient is a substance or product used in the production or manufacture of a food, including flavourings, food additives and food enzymes, as well as parts of the compound ingredient found in the finished product as such or in some other form. All ingredients shall be listed in descending order of weight, as recorded at the time of their use in the manufacture of the food. Added water and volatile ingredients shall be listed in order of their weight in the finished product.
- Vitamins and minerals shall be indicated in the labelling using the names listed in Annex XIII to the Food Information Regulation (Table 1). Listing nutrients on the list of ingredients and on the list of characteristic substances in a uniform manner ensures that it is easier for the consumer to identify the ingredients of the product. The name of the nutrient can also be supplemented with the chemical name of the nutrient compound, e.g. vitamin C (I-Ascorbic Acid) However, authorised novel food ingredients shall be indicated on the list of ingredients of the food supplement in the form indicated in the conditions of their use. Authorised novel foods and their conditions of use are listed on the Union list (EU) 2017/2470 (latest consolidated version).
- The Finnish Food Authority recommends the use of the Finnish names of plants in the list of ingredients, if available (for example, using the Finnish name "pakurikääpä" for Inonotus obliquus fungus instead of the name "chaga" which is of Russian origin), or some other names established in Finland (for example, the Lycium barbarum plant can be referred to by the Finnish name "pukinpensas" or the established name "goji"). If no Finnish name is available, it is advisable to use the full scientific name of the plant (plant family + species, e.g. Griffonia simplicifolia).
- Ingredients causing allergies and intolerances, and preparations made out of them, shall always be indicated, even when they have not been used as ingredients in the food product as such, but have ended up in the food product in some other way (e.g. additive carriers). In addition to this, these ingredients shall be emphasised on the list of ingredients by using e.g. a different font size, style or background colour. Mandatory indication applies to the ingredients and products causing allergies and intolerances listed in Annex II of the Food Information Regulation.
- Additives shall be indicated on the list of ingredients by the name of the category indicating their purpose of use, supplemented by their own name or number code (E code). Substances causing allergies and intolerance shall be indicated with emphasis in the E numbers of the additives (e.g. emulsifier E 322 (soya) or in the name of the additive (e.g. emulsifier (soy lecithin).
- The quantity of the ingredient must be indicated if it is an essential ingredient for the characterisation of the food (see section 4.3 The amount of characteristic substances of the food supplement in the daily dose), if the ingredient is present in the name of the food or otherwise emphasised on the labelling, e.g. in words or images.
- “Best before” or “Use by” date
- If the date of minimum durability is not indicated to the nearest day and month, the following form of indication shall be used: "Best before end…" (e.g. "Best before end 12/2025").
- Country of origin or place of provenance of a food or, if necessary, its primary ingredient as provided for in the Food Information Regulation or in provisions issued by virtue of it, or in other provisions.
- Instructions for use (see also Section 4.3)
- Warnings are also part of instructions for use.
- Food supplement operators shall establish whether the products they bring to market are suited to all consumers, or whether warnings or restrictions of use should be provided on the labelling (For example, the product is not intended for children under the age of 10).
- NOTE: The warning label required for food supplements that states the product must be kept out of the reach of small children is a storage instruction and not an instruction for use. In addition to it, where necessary, the food supplement must include instructions on who the product is intended for or who should avoid its use.
- Any special storage conditions and/or conditions of use, if necessary
- The alcohol content of liquid food (for solid foods on a voluntary basis), if necessary
- The code of the food batch (Decree of Ministry of Agriculture and Forestry 834/2014, section 5)
- Instead of the food batch code, an indication based on the date of minimum durability, or the use-by date can be used, provided it is given as an indication of at least the day and the month (Figure 3).
As opposed to other foods, the requirement for a nutrition declaration set out in the Food Information Regulation is not applied to food supplements. Therefore, the information provided on food supplements does not include a nutrition declaration, but the amounts of characteristic substances in the food supplement are indicated for the recommended daily dose (see section 4.3).
The Finnish Food Authority recommends that, for food supplements designed to be stored and used with inner packaging, the labelling provided on the inner packaging should contain indications that are necessary to protect the health and the financial interests of the user, such as “name of food product”, “Best before” date / “Use by” date, any required storage conditions and instructions for use (for example, the recommended daily dose and any warnings).
The size of the labelling used on foods, including food supplements, is provided for in the Food Information Regulation. Mandatory particulars for food supplements shall be printed on the packaging or on the label using a font size where the x-height is equal to or greater than 1.2 mm. On small packaging, whose largest surface has an area of less than 80 cm2, the x-height shall be equal to or greater than 0.9 mm. If the mandatory particulars are printed in capital letters, the x-height shall be 1.2 mm.
More information about the general labelling provisions can be found in the Finnish Food Authority’s Food Information Guide for food control authorities and food business operators (in Finnish and Swedish only). (Finnish Food Authority guideline 17068) and the Finnish Food Authority website (in Finnish and Swedish only).
4.3 Labelling pursuant to the Decree on Food Supplements
According to the Decree on Food Supplements, the labelling of food supplements must provide the following information in addition to general labelling information:
- The trade name “food supplement”
- The names of the categories of nutrients or substances that characterise the product or an indication of the nature of those nutrients or substances
- A category of characteristic nutrients or substances refers to the group in which the characteristic substance of the food supplement can be classified. Such categories include vitamins, minerals, fibres, flavonoids, amino acids, fatty acids or plant or herbal extracts. The category or the nature can be expressed in the food product’s name (e.g. lactic acid bacteria preparation or chewable vitamin tablet).
- The amount of characteristic substances of the food supplement in the daily dose
- The amounts of the characteristic substances of the food supplement must be indicated on the labelling in numerical form as the daily dose of the product recommended by the manufacturer. The units used for vitamins and minerals are stipulated in Annex I of the Food Supplements Directive and Table 1 of this Guide. The values given are averages based on an analysis of the product made by the manufacturer. The capsule or binding agent of the preparation must be taken into account in the indicated nutrient amount. The amount of the characteristic substances of the food supplement must always be expressed as percentage of the daily reference intake, if the reference intake is available for the substance in question. The reference intakes of vitamins and minerals are presented in Annex XIII to the Food Information Regulation.
- In the case of vitamins and minerals, the vitamin activity of the compounds must also be taken into account and indicated for the characteristic substances as the proportion with physiological activity (e.g. thiamine hydrochloride must be indicated as free-form thiamine). Carotenoid equivalence factors (indicating vitamin A) can be found on the Finnish Food Authority website.
- For vitamins and minerals contained in food supplements, the tolerances indicated in Table 3 are permitted; they include the uncertainty of measurement associated with a measured value. If claims are used in the marketing of a food supplement, the content of the vitamin or mineral referred to in the claim may not be less than the indicated amount by more than the measurement uncertainty of the analytical method. The tolerances are based on the EU Guidance on Tolerances (pdf).
- No specific tolerances have been given for other characteristic substances in food supplements. According to the Decree on Food Supplements, the amount of characteristic substances must be an average based on the manufacturer’s analysis of the product. The amount of the characteristic substance can therefore be regarded, on one hand, as the minimum amount which the manufacturer guarantees the food supplement will contain until the “Best before” or “Use by” date, and, on the other, as the maximum daily dose set by the manufacturer that must not be exceeded. Therefore, because the amount of the characteristic substance indicated by the manufacturer should be present in the food supplement, the Finnish Food Authority holds that only the measurement uncertainty of the analytical method is acceptable as a tolerance from other characteristic substances in food supplements. If the amount indicated on the packaging differs from the amount found in the analysis by more than the measurement uncertainty, this leads to an obligation on the operator to determine the cause of the deviation and to take the necessary corrective measures.
When the analysis results are repeatedly at the limits of the permitted tolerance, the operator should intensify self-monitoring and make the necessary changes to the production process or to the information provided on the food supplement.
Food supplements in which the declared concentrations of the characteristic substances are repeatedly different from the stated tolerance limits are not acceptable and should not be marketed.
Additional information on permitted nutrient tolerances can be found on the Finnish Food Authority website.
- Recommended daily dose
- Warning that the recommended daily dose indicated must not be exceeded
- Statement that a food supplement should not be used as a substitute for a varied diet
- Warning that the product must be stored out of the reach of small children.
- NOTE: this is a storage instruction, not an instruction for use (see also section 4.2). In addition, where necessary, the food supplement must include instructions on who the product is intended for or who should avoid its use.
Table 4. Permitted tolerances of nutrients in food supplements (including measurement uncertainty) when no nutrition or health claims are used.
| Nutrient | Permitted tolerances of nutrients (including measurement uncertainty) in food supplements |
|---|---|
| Vitamins | +50 % ** -20 % |
| Minerals | +45 % - 20 % |
** for vitamin C in liquids, higher upper tolerance values can be accepted.

Figure 2. Example of food supplement labelling
4.4 Other labelling provisions
Information on labelling provisions based on other acts and regulations shall also be taken into account in the labelling of food supplements, which is described in greater detail below.
4.4.1 Genetically modified ingredients
Regulation (EC) No. 1829/2003 of the European Parliament and of the Council on genetically modified food and feed stipulates that consumers shall be informed of any genetically modified ingredients used in a food supplement in the EU. The term "genetically modified" or, for example, "produced from genetically modified (ingredient name)", shall appear in the labelling in the list of ingredients immediately following the ingredient concerned.
Additional information on the labelling of genetically modified products can be found on the Finnish Food Authority website.
4.4.2 Irradiation
If the dried spice herbs, spices or spice plants contained in a food supplement have been irradiated to improve their microbiological quality, this must be indicated in the labelling (Decree 852/2000 of the Ministry of Trade and Industry on treatment of food with ionizing radiation). Irradiation may only be carried out in facilities approved by the European Union. The irradiation of other ingredients is not permitted and no food supplements containing other irradiated ingredients may be placed on the market. More information on the irradiation of foodstuffs can be found on the Finnish Food Authority website (in Finnish and Swedish only).
NOTE: If the ingredient has not been irradiated at a facility approved by the EU, or if the food supplement contains an irradiated ingredient for which irradiation is not permitted, this constitutes a major error, which requires immediate withdrawal of the product from the market.
4.4.3 Labelling referring to an organic production method
Labelling referring to an organic production method may only be used provided the operator is included within the scope of a control system as referred to in Regulation (EU) 2018/848 of the European Parliament and of the Council on organic production and labelling of organic products.
The labelling of a product is considered to refer to an organic production method, if the product and/or an ingredient of the product is in labelling, advertising material and/or commercial documents described using phrases suggesting to the purchaser that the product and/or its ingredients have been produced in accordance with provisions pertaining to organic production.
More detailed information on labelling referring to an organic production method can be found in the Finnish Food Authority guideline General terms and conditions for Organic Production (in Finnish and Swedish only).
4.4.4 Labelling referring to the use of sweeteners
Sweeteners authorised in food supplements are listed in Part E of Annex II to the Food Additives Regulation (under food group 17). If sweeteners are used in food supplements, their use shall also be indicated on the package of the food supplement as follows:
- If the food supplement contains sweeteners, the statement “with sweetener(s)” shall be included in or accompany the name of the food supplement.
- If the food supplement contains both sugar and sweeteners, the statement “with sugar(s) and sweetener(s)” shall be included in or accompany the name of the food supplement.
- If the food supplement is sweetened with aspartame/aspartame-acesulfame salt, the statement “contains aspartame (a source of phenylalanine)” shall appear on the label in cases where aspartame/aspartame-acesulfame salt is designated in the list of ingredients only by reference to the E number. The statement “contains a source of phenylalanine” shall appear on the label in cases where aspartame/aspartame-acesulfame salt is designated in the list of ingredients by its specific name.
- If the recommended daily dose of the food supplement contains more than 10% of added polyols, the statement "excessive consumption may produce laxative effects" shall be provided on the packaging.
4.4.5 Lactose-free and gluten-free products
If the product is marketed as lactose-free or gluten-free, the product shall meet the criteria related to these claims.
Read more about lactose-free and gluten-free -claims on the Finnish Food Authority website.
4.4.6 Labelling referring to caffeine
If the food supplement contains, as a characteristic substance, caffeine or an ingredient containing caffeine, the statement “Contains caffeine. Not recommended for children or pregnant women” shall appear on the label. The statement shall be placed in the same field of vision as the name of the food The statement shall be followed by a reference in brackets to the caffeine content expressed in mg per recommended daily dose.
Additional information on warnings and instructions for use for foods containing caffeine can be found on the Finnish Food Authority website.
4.5 Marketing
According to Article 7 of the Food Information Regulation, truthful and sufficient food information shall be provided. Food information shall not be misleading. This applies to all commercial communications regarding the food, including websites, social media, network marketing and word of mouth marketing, as well as the appearance and presentation of the food.
The Consumer Protection Act (38/1978) also applies to the marketing of food products. The Act prohibits marketing procedures that violate good practice or are otherwise inappropriate from a consumer standpoint. Marketing that does not contain information necessary to the health or financial security of consumers is always inappropriate. The Consumer Ombudsman supervises marketing and customer relations practices from a consumer protection standpoint.
Regulation (EC) No. 1924/2006 on nutrition and health claims made on foods (hereinafter Claims Regulation) stipulates on nutrition and health claims used in the packaging, presentation and advertising of foodstuffs.
More detailed guidelines and interpretations concerning the use of nutrition and health claims (in Finnish and Swedish only) in the promotion of foodstuffs can be found on the Finnish Food Authority website.
4.5.1 Nutrition claims
According to the Claims Regulation, a nutrition claim is any claim which states, suggests or implies that a food has particular beneficial nutritional properties with regard to nutrients or other substances it contains, contains in reduced or increased proportions, or does not contain.
Only the health claims listed in the Annex to the Regulation may be used, and then only if they conform to the requirements specified in the Regulation. The specification for nutrition claims states how much a given nutrient must be found in a given food in order for use of the nutrition claim to be authorised. In the case of food supplements, the conditions of use must be met in the recommended daily dose of the food supplement.
Nutrition claims may concern e.g. energy, amino acids, fat and nutritional fibre as well as vitamins and minerals. If such claims are made on a food supplement, the food business operator must have evidence to substantiate the claims.
Nutritional claims include:
- high-fibre or a source of fibre
- high vitamin or mineral content
- a source of omega-3 fatty acids.
Additional information on nutrition claims can be found on the Finnish Food Authority website.
4.5.2 Health claims
According to the Claims Regulation, a health claim is any claim that states, suggests or implies that there is a relationship between a food category, a food, or one of its ingredients and health. A pictorial, graphic or symbolic representation in any form can also be considered a claim.
Health claims include:
- Calcium is needed for the maintenance of normal bones
- Iodine contributes to normal energy-yielding metabolism
- Vitamin C contributes to the reduction of tiredness and fatigue
- Glucomannan in the context of an energy restricted diet contributes to weight loss
- Calcium and vitamin D help to reduce the loss of bone mineral in post-menopausal women. Low bone mineral density is a risk factor for osteoporotic bone fractures.
Only authorised health claims included on the list of authorised health claims may be used in the marketing of foods, provided the product meets the criteria defined for the use of health claims with respect to, among others, composition.
The EU Register of Health Claims can be found on the European Commission website.
For substances of plant origin, only those health claims may currently be used on which the scientific assessment procedure of the European Food Safety Authority (EFSA) is still ongoing; they may be used until the assessment procedure has been completed and the Standing Committee of the European Commission has made a decision to either authorise or reject them. The use of all other claims is prohibited.
Health claims require the inclusion of certain additional information in the labelling or, if no such labelling exists, in presentation and advertising. These are:
- the daily dose amount of the substance referred to in the claim
- a statement indicating the importance of a varied and balanced diet and a healthy lifestyle*
- information on how much and how often the food should be consumed in order to obtain the claimed beneficial effect
- where appropriate, a statement addressed to persons who should avoid using the food, and an appropriate warning for products that are likely to pose a health risk if consumed in excess
- when a claim referring to a reduction in the risk of disease is used, the following statement shall be added: “The disease to which the claim is referring has multiple risk factors and altering one of these may or may not have a beneficial effect.”
* As the statement “a food supplement must not be used as a substitute for a balanced diet” is also required under the Decree on food supplements, the Finnish Food Authority holds that the mandatory labelling according to both the Claims Regulation and the Decree on food supplements can be combined with the following sentence: “A food supplement is not a substitute for a varied and balanced diet or a healthy lifestyle.”
Additional information on health claims can be found on the Finnish Food Authority website.
4.5.3 Medicinal claims
The Food Information Regulation and the Decree on food supplements both stipulate that properties relating to the prevention, treatment or curing of human diseases must not be presented or referred to in the labelling, brochures or advertising of food supplements or in any other way.
The product effects must not give a medicinal impression, such as by using medical terminology or referring to changes, symptoms, ailments or pains caused by diseases in such a way that marketing attributes properties relating to the prevention, treatment or curing of diseases to the food product.
Only medicinal products, herbal medicinal products and traditional herbal medicinal products as well as homeopathic and anthroposophic preparations may be presented as having medicinal purposes. The sale of these products requires applying to Fimea for marketing authorisation or registration, which assesses, for example, the quality, effectiveness and safety of the product.
Examples of claims that may not be used on food supplements
- protects against bacteria and viruses
- helps those suffering from hypertension
- Mrs. X's arteriosclerosis disappeared when she used preparation Y
- for anaemia
- using the word medicine, medicinal herb, or drug in the name of the food supplement (also in other languages).
4.5.4 Other prohibited claims
Under both the Decree on food supplements and the Claims Regulation, the labelling, presentation and advertising of a food supplement may not present or suggest that a balanced and varied diet does not generally provide enough nutrients.
Under the Claims Regulation, the following claims, among others, are also prohibited:
- claims making reference to recommendations of individual doctors or health professionals
- claims making reference to the rate or amount of weight loss
- claims suggesting that health could be affected by not consuming the product
- claims giving rise to doubt about the safety or adequacy of other foods
- claims encouraging excess consumption of the product
- claims making reference to changes in bodily functions which could give rise to fear in consumers.
Figure 3. Example of food supplement labelling with additional labelling (legislative basis for different labelling requirements expressed by references
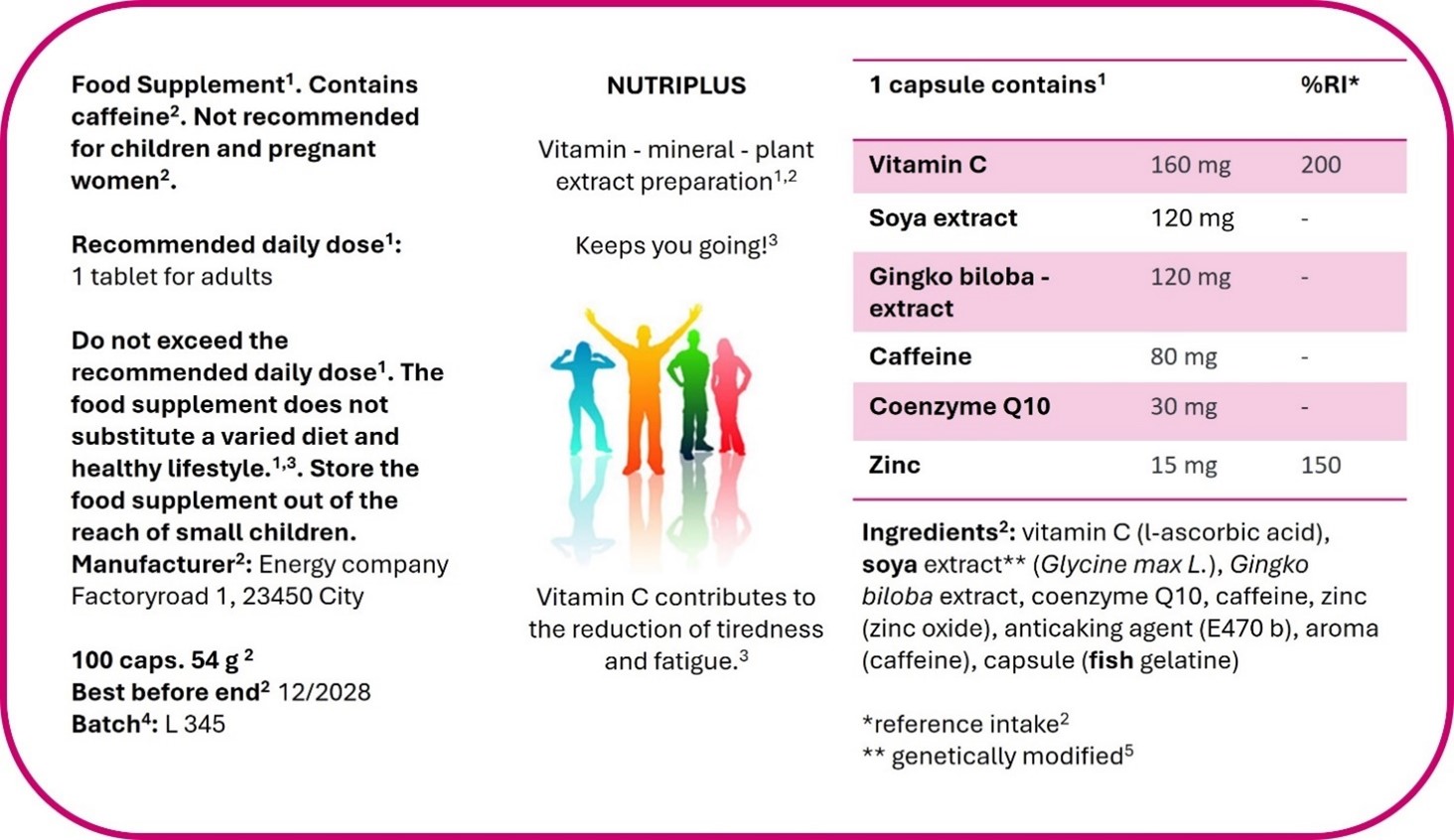
1) Regulation on food supplements 78/2010, including sales names and the amount of substances that characterise food supplements in the recommended daily dose.
2) Regulation (EU) No 1169/2011, including list of ingredients in descending order with allergens emphasized, company contact details, caffeine labelling.
3) Regulation (EC) No 1924/2006, including general health claims and claims specifying them, additional labelling required by the claims.
4) Regulation 834/2014 on the provision of food information to the consumer: batch number.
5) Regulation (EC) No 1829/2003 on genetically modified food and feed: GM labelling.
5 Food supplement interfaces
Food supplements are foods that differ from other food categories in terms of their physical form and intended use. Food supplements may be very similar to some other food category, making it difficult to distinguish between different products. Food supplements can also resemble medicinal products in their form and intended use, in some cases even in their composition. Interfaces between food supplements and other products are discussed below.
5.1 Differentiation from conventional food products
Food supplements are foods that differ primarily from other food categories in terms of their physical form and intended use. However, with some products it is more challenging to differentiate, as a food supplement and a conventional food can be identical in terms of, for example, their form.
Fibre preparations are an example of this - they can be marketed as conventional foods to be added to, for example, porridge or yoghurt in a desired amount or according to the instructions for use. A fibre preparation can also be marketed as a food supplement for use as a concentrated source of fibre, if it is to be consumed in small unit quantities. In this case, the fibre product must provide at least 3 mg of fibre in the recommended daily dose of the food supplement (source of fibre is a condition for using a nutrition claim).
The food category of the product is thus determined based on its intended use, i.e. the purpose for which the food is marketed. A product that meets the criteria defined for a food supplement (see Section 2 Definition of food supplement) can be placed on the market as a food supplement, provided the labelling information referred to in regulations pertaining to food supplements is provided on the product and a food supplement notification has been submitted to the Finnish Food Authority regarding the product (see Section 6.2).
5.1.1 Classification of confectionery and chewing gums
As a rule, the Finnish Food Authority considers confectionery and chewing gums to be conventional foods, or possibly fortified foods if vitamins or minerals have been added to them. However, based on their form, confectionery and chewing gums also meet the definition presented in the Decree on food supplements and they can be classified as food supplements if their intended use is clearly characteristic of that of food supplements. Factors supporting classification as a food supplement also include, for example, a container provided with a child-proof lid or the product being clearly marketed as a food supplement and not as confectionery or a chewing gum. For example, the name of a food supplement cannot contain references to confectionery, such as the words “candy” or “sweet”. Other factors supporting classification of the product as a food supplement might be, for example, selling the product in a jar fitted with a safety cap and clearly marketing the product as a food supplement, not as confectionery or chewing gum.
5.1.2 Products marketed to athletes
Depending on their content, formulation and intended use, foods marketed to athletes are either
- conventional foods (e.g. whey protein, cottage cheese, quark, smoothie)
- fortified foods (e.g. sports nutrition powders, bars, drinks fortified with vitamins and minerals)
- food supplements (e.g. capsules, tablets, low-energy powders consumed in small unit quantities).
Protein, amino acid and carbohydrate powders, bars and drinks marketed to athletes are in most cases conventional or fortified foods. Some foods marketed to athletes are, however, classified as food supplements based on their intended use and method of use as well as their energy content. Sports nutrition products classified as food supplements differ from other food products in their formulation, i.e. they come in pills, capsules, powders or herb extracts, among others. Products intended for use as a source of, for example, vitamins, minerals, amino acids, fibre and fatty acids as well as various plant extract preparations are food supplements. What is essential is that the amount of energy from food supplements is low, whereas conventional foods for athletes can contain high levels of energy and can even be used as a substitute for a meal or a part of a meal. Because food supplements are consumed in small unit quantities, drink-type products designed for athletes do not meet the definition of a food supplement. This also applies to drink powders, which intended use is for making drinks. On the other hand, effervescent tablets and powders dissolved in a small amount of liquid (approximately 1-2 dl) to facilitate ingestion of the product can be considered food supplements.
The Finnish Food Authority holds that the energy provided by a food supplement when consumed according to its recommended maximum dosage is not significant in terms of energy intake, provided that it does not exceed 200 kJ (50 kcal) per day. A teaspoon or tablespoon (5-15 ml) of the marketed product is considered a small dose, but the daily dose of the food supplement must not exceed 100 ml by volume.
5.2 Differentiation from fortified food products
At present, the distinction between food supplements and fortified foods is not fully clear in every respect, and no common guidelines have been issued at the EU level. Fortified foods are foods to which nutrients, most commonly vitamins or minerals, have been added. Fortified foods and food supplements share the same intended use: to supplement the normal diet.
However, in terms of their physical form, fortified foods are more similar to conventional foods than to food supplements. Fortified foods include, for example:
- milk fortified with vitamin D
- soft drinks fortified with vitamins and minerals
- energy drinks containing caffeine/guarana and vitamins of group B
- margarine supplemented with vitamins A and D
- breakfast cereals fortified with minerals
- iodised salt
- meal replacements for weight control.
Fortified foods are also consumed in larger quantities than food supplements, and the amount of energy they yield often has a significant impact on the diet. Food supplements, on the other hand, are consumed in small unit quantities and are not intended to provide a significant amount of energy.
Additional information on fortified foods can be found on the Finnish Food Authority website.
5.3 Differentiation from foods for specific groups
Foods for specific groups include:
- infant formulae and follow-on formulae
- cereal-based processed foods and baby foods
- foods for special medical purposes
- total diet replacements for weight control.
Foods for specific groups may be the only source of nutrition for their users, replacing every meal of the day, or they can be used as supplementary foods, providing part of the daily dietary needs.
As a rule, foods for specific groups do not come in pill or capsule form. A nutritionally incomplete or nutritionally adapted food for special medical purposes that does not serve as the only food source may also be a product that comes in pill or capsule form.
A food for special medical purposes is a product intended for the dietary management of patients and should be used under medical supervision. It is intended for patients, whose ability to properly consume, digest, absorb, metabolise or excrete normal foods or certain nutrients or metabolites contained therein is insufficient, deteriorated, or impaired, or for patients with other medically determined nutrient requirements whose dietary management cannot be achieved by modification of the normal diet alone.
If dietary intake can be secured by changing a normal diet or by using a food supplement, the product is not considered a food for special medical purposes. A product that does not contain nutrients is also not a food for special medical purposes. The use of nutrition and health claims is not permitted for foods for special medical purposes. However, the prohibition does not preclude the dissemination of any useful information or recommendations on the product to persons with qualifications in medicine, nutrition, pharmaceutics, or for other healthcare professionals responsible for maternal care and childcare.
Additional information on foods for specific groups can be found on the Finnish Food Authority website.
5.3.1 Food supplements for children
Food supplements intended for children are not baby foods but are primarily subject to the same legislation as food supplements intended for adults. For example, the daily reference intake is the same in food supplements for both adults and children. However, the lower tolerable upper intake levels (UL) of nutrients must be taken into account in food supplements intended for children. In other words, the nutrient content should be lower in daily doses of food supplements intended for children than in similar food supplements for adults. The nutrient content in food supplements intended for the whole family must also be such that the UL values are not exceeded for any age group.
It should also be noted that additives may not be used in food supplements intended for children under three years of age. This is because the Food Additives Regulation does not provide a list of permitted food supplement additives for this age group.
In addition, the use of some authorised novel foods in food supplements is not permitted in food supplements intended for infants and young children. The conditions of use for each authorised novel food are listed in the Union list of authorised novel foods (EU) 2017/2470.
Nutrition claims may be made in the marketing of food supplements intended for children. Authorised health claims compliant with Article 14(1b) of the Claims Regulation may also be made if the food supplement meets the conditions of use of the claim. No other health claims may be made.
5.4 Differentiation from medicinal products
Food supplements may in some cases contain substances or herbal medicinal products from the list of medicines (e.g. Echinacea). In this case, the use must be based on something other than the medicinal effect of the product or the substance or plant it contains (see Section 2 Definition of food supplement). In other words, there may be substances on the market that are identical or products that are made from the same plants, but different requirements are applied to their manufacture and marketing depending on whether they are placed on the market in accordance with regulations on medicinal products or food regulations.
If a food business operator intends to place on the market a food supplement containing a substance or herbal medicinal product found on the list of medicines, the Finnish Food Authority recommends that the operator contact Fimea to assess the need for classification. Fimea may classify a product as a medicine also at its own discretion if the product meets the criteria defined for a medicine. The list of medicines can be found on the Fimea website.
5.4.1 Classification of medicinal products
In accordance with section 3 of the Medicines Act (395/1987), a medicinal product is a preparation or substance intended for internal or external use to cure, alleviate or prevent a disease or its symptoms in humans or animals. Preparations or substances to be taken internally or externally for the purposes of determining the state of health or the cause of a disease, or for restoring, correcting or modifying physiological functions in humans or animals are also considered to be medicinal products. Products marketed as medicinal products must always have a marketing authorisation from or be registered by Fimea.
Under the Medicines Act, Fimea is the authority who decides, when necessary, whether a substance or preparation is to be considered a medicinal product. Each preparation is classified individually. Products are classified into medicinal products or non-medicinal products. The classification is based both on the composition and the effect of the preparation, as well as on its intended use. Products used in accordance with the definition of a medicinal product (section 3 of the Medicines Act) have a medicinal use and are thus considered medicinal products. Instructions for the filing of a classification application can be found on the Fimea website.
Products classified as medicinal products may not be marketed as foods. The operator is responsible for the marketing of their product and for the selection of the correct sales channel as well as for withdrawing any products classified as medicinal products from the market.
5.4.2 List of medicines
Fimea has confirmed and issued a decision on the List of medicines, which includes three appendices. The decision includes a list of the substances and herbals that are in medicinal use in Finland (Appendices 1 and 2), as well as a list of medicinal substance analogues and prohormones (Appendix 1 A), which based on their effect are always considered comparable to medicinal products supplied by prescription only. The list of medicines is not exhaustive. All substances and herbals that meet the definition of a medicinal product under the Medicines Act are considered medicinal products.
According to the decision, medicinal products include: the substances listed in Appendix 1 to the List of medicines; and the salts and esters of those substances; the doping substances listed in the valid Government Decree on doping substances as referred to in chapter 44(16)(1) of the Criminal Code of Finland; and the substances listed in Appendix 1 A.
Medicinal products also comprise the following substances or products insofar as they are used in accordance with section 3 of the Medicines Act:
- the herbal medicinal products listed in Appendix 2 to Fimea's decision, and the active substances derived from them, which include herbal medicinal preparations (e.g. extracts, tinctures, pressed juices) and comparable active substances of animal origin, as well as medicinal products manufactured from them.
- certain products or substances that deviate from normal medicinal products in form, composition, method of manufacture or mechanism of action, such as radioactive medicinal products, allergen products, vaccines and medicinal gases, medicinal products used in advanced therapies as well as medicinal products derived from human blood and plasma.
- and vitamins and mineral products.
5.4.3 Herbal medicinal products and homeopathic preparations
Herbal medicinal products and homeopathic preparations are divided into herbal medicinal products subject to marketing authorisation, registered traditional herbal medicinal products and registered homeopathic preparations. Anthroposophic products that meet the definition of a homeopathic preparation in the Medicines Act are also considered homeopathic preparations. Fimea controls all herbal medicinal products.
Herbal medicinal products are medicinal products, whose active ingredients contain herbal substances or herbal preparations or a combination of these.
A traditional herbal medicinal substance refers to a medicinal product for human use that contains substances of plant origin, herbal medicinal products or a combination of these as its active ingredients. In addition, it must fulfil the criteria for registration laid down in section 22 (1) of the Medicines Act. A traditional herbal medicinal product may also contain vitamins or minerals if they contribute to the effect of active substances of plant origin.
A homeopathic preparation as a medicinal product that has been manufactured from homeopathic stocks using the homeopathic manufacturing procedure described in the European Pharmacopoeia. Homeopathic and anthroposophic preparations may be subject to marketing authorisation or registration.
The labelling of a food supplement is distinctly different from that of an herbal medicinal product (see Figure 5). The labelling on herbal medicinal products is determined on the basis of the Medicines Act. For example, the labelling of a traditional herbal medicinal product shall contain the following statements in Finnish and Swedish: ”PERINTEINEN KASVIROHDOSVALMISTE – TRADITIONELLT VÄXTBASERAT LÄKEMEDEL” (“TRADITIONAL HERBAL MEDICINAL PRODUCT”) and ”Ota yhteys lääkäriin, jos oireet jatkuvat tai jos hoidon aikana esiintyy haittavaikutuksia / Kontakta läkare om symtomen fortsätter eller om det förekommer biverkningar under behandlingen” (“Consult a physician if the symptoms persist or if adverse effects occur during the use of the product”). The labelling shall also indicate the registration number (R xxx FIN)” and Nordic Product Number (Vnr xx xx xx).
The labelling of homeopathic and anthroposophic preparations, both subject to marketing authorisation and registration, shall include the following statement in Finnish and in Swedish: ”HOMEOPAATTINEN VALMISTE – HOMEOPATISKT MEDEL” (“HOMEOPATHIC PREPARATION”) or ”ANTROPOSOFINEN VALMISTE – ANTROPOSOFISKT MEDEL” (“ANTHROPOSOPHIC PREPARATION”) and the registration number (H xxx FIN, which is, if necessary, followed by the degree of dilution, e.g. D6).
Additional information on herbal medicinal products and homeopathic preparations can be found on the Fimea website.
5.4.4 Sale and marketing of medicines and foodstuffs in pharmacies
Fimea controls medicinal products, their manufacture, distribution, marketing and sale. Medicinal products subject to marketing authorisation may only be sold to the public from pharmacies, subsidiary pharmacies, pharmacy service points or online pharmacies. With regard to herbal medicinal products and homeopathic or anthroposophic preparations, a grocery store may also serve as a sales outlet. Fimea determines the sales outlets for products in connection with the granting of a marketing authorisation or registration.
In order to clearly differentiate between food and medicinal products and to eliminate the possibility of misunderstanding among consumers, the Finnish Food Authority holds that medicinal products and food must be clearly differentiated from each other in advertising. They must not be marketed in a misleading manner using the same materials. According to Article 7 of the Food Information Regulation, food information must be truthful and sufficient in food packaging, presentation and advertising, or in some other way in connection with marketing. It is also prohibited to provide misleading information on foods. Article 16 of Regulation (EC) No. 178/2002 of the European Parliament and of the Council laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety also includes provisions on the prohibition of misleading information. Section 25b of the Decree on Medicines (693/1987) also stipulates that in marketing medicinal products to the public, advertisements must make it clear that they are advertisements marketing medicinal products.
More information on food display, product grouping and marketing in stores and pharmacies can be found in the Food Authority's Nutrition and Health Claim Guide (paragraph 7.2) (in Finnish and Swedish only).
Table 5. Requirements and controls for placing foodstuffs and medicinal products on the market
| Foodstuffs | Medicinal products | |||
|---|---|---|---|---|
| Products | Conventional foods | Novel foods | Food supplements Fortified foods Infant formulae Certain follow-on formulae Foods for special medical purposes Total diet replacements for weight control |
Medicinal preparations: Medicinal products subject to marketing authorisation, including herbal medicinal products Registered traditional herbal medicinal products Homeopathic and anthroposophic preparations |
| Criteria for entering the market | No specific authorisation or notification requirements for placing on the market | Authorisation procedure | Notification procedure | Authorisation procedure (marketing authorisation or registration) |
| Authorisation or notification authority | - | European Commission | Finnish Food Authority | Fimea European Medicines Agency |
| Control |
|
Fimea | ||
5.5 Differentiation from medical devices
According to Regulation (EU) 2017/745 of the European Parliament and of the Council on medical devices, “’medical device’ means any instrument, apparatus, appliance, software, implant, reagent, material or other article intended by the manufacturer to be used, alone or in combination, for human beings for one or more of the following specific medical purposes:
a) diagnosis, prevention, monitoring, prediction, prognosis, treatment or alleviation of disease,
b) diagnosis, monitoring, treatment, alleviation of, or compensation for, an injury or disability,
c) investigation, replacement or modification of the anatomy or of a physiological or pathological process or state,
d) providing information by means of in vitro examination of specimens derived from the human body, including organ, blood and tissue donations,
and which does not achieve its principal intended action by pharmacological, immunological or metabolic means, in or on the human body, but which may be assisted in its function by such means.
The following products shall also be deemed to be medical devices:
e) devices for the control or support of conception; and
f) products specifically intended for the cleaning, disinfection or sterilisation of devices.”
Devices and supplies for health care purposes shall be designed, manufactured, packaged and labelled in such a manner that they are suitable for the purpose intended by the manufacturer. The manufacturer is responsible for ensuring that the device:
- is in conformity with the definitions concerning it
- meets the general safety and performance requirements
- does not jeopardise the safety of the patient, the user or any other person.
The manufacturer must specify the intended use of the product as referred to in the definition of health care equipment and supplies. Health care equipment and supplies placed on the market shall be given a CE marking, with the exception of devices manufactured for personal use and investigational devices with no CE marking. The CE marking constitutes the manufacturer's warranty that the equipment or supplies meet the essential requirements applied to them.
In some cases, a preparation for oral consumption may also be deemed a device, if its intended use and mechanism meet the criteria of health care equipment and it fulfils the essential requirements applied to the device. Medical devices are controlled by Fimea.
Additional information on medical devices can be found on the Fimea website.
6 Notifications required of food business operators
6.1 Notification of a food business
Activities related to food supplements, such as manufacture, contract manufacture, import, export and sales, are food-related activities that must be notified to the food control authority of one’s own municipality. The composition of food supplements and the information provided on them involve a wide range of risks, which is why notifications are even required for the small-scale sale, marketing or conveying of food supplements. A notification of the business must be submitted even if the seller does not actually have any food supplements in their physical possession and they only market or convey them, or if they do not maintain food premises. Such businesses include agency operations and different types of distance selling, such as the online, telephone, mail order and network marketing of food supplements (Article 15 5) to the Regulation (EU) 2017/625 of the European Parliament and of the Council on official controls) If a company’s operations are expanded to include distance selling, this must be reported to the municipal food control authority.
A notification of a food business for the registration of operations must be submitted to the food control authority of one’s own municipality no later than four weeks prior to the start of the business operations. The notification may be submitted online using the Ilppa service (in Finnish and Swedish only) or by completing the notification form found on the website of the municipality. The notification referred to in Section 10 of the Food Act must also be submitted, if essential changes take place in the business. Likewise, a notification must be submitted to the municipal food control authorities in the event of any interruption or cessation of operations or a change of operator. The processing of the notification is subject to a fee in accordance with the municipality’s price list.
Food businesses handling products of animal origin or raw materials of animal origin before retail sale, shall apply for approval of the premises from the municipal food control authorities before the beginning of operations or any significant change in operations. In connection with the application, an own-check plan must be presented to the food control authority (see section 7.1).
Read more about setting up a food business on the Finnish Food Authority website.
6.2 Food supplement notification
According to Section 6 of the Food Act, a food business operator who manufactures, has a food supplement manufactured, imports (from the internal market or third countries) or otherwise places on the Finnish market a food supplement, shall submit a notification to the Finnish Food Authority.
The Finnish Food Authority considers that the operator responsible for placing the food supplement on the market should submit a food supplement notification by no later than when the product is placed on the market. A notification is also required when the composition of the food supplement with respect to its characteristic substances changes. No notification of food supplement exiting from the market is required.
A notification is also required in the following cases:
1. The same food supplement is placed on the market by several different operators
- If a food supplement has several responsible operators (e.g. several importers who sell the food supplement under their own name and are thus responsible for the food supplement information given in Finland), each of these must submit a notification for the food supplement in question. The information given in the notification and on the labelling must be consistent.
2. Change of the operator responsible for placing the food supplement on the market
- The notification shall also be made whenever the operator responsible for placing a food supplement on the market changes. Even though the food business operator previously responsible for a food supplement had submitted the required notifications for it, this food supplement is now considered new for the new food supplement operator and a notification for it must be submitted. If, on the other hand, the operator responsible for a food supplement remains the same (the business ID does not change), but the name of the operator changes, no new notifications are required.
3. The name of the food supplement changes significantly
- If the name of a notified food supplement changes in such a way that the average consumer can no longer identify the food supplement as the same product, the food supplement in question is considered new and a notification for it must be submitted. If the name of a notified food supplement changes only slightly, so that the average consumer can still identify the food supplement as the same product, the change is considered so insignificant that no new notification is required.
According to section 7 of the Decree on food supplements, a model of the labelling used on the product must be attached to the food supplement notification. The model must show both statutory and voluntary labelling and, where possible, images planned for the packaging. If the signatory of the notification is a party outside the notifying business, such as a consulting company in the sector, a power of attorney must also be attached. Other appendices to the notification may include, for example, a product brochure.
The food supplement notification is submitted via the Finnish Food Authority’s electronic service. The notification may be submitted in Finnish, Swedish or English. Log in to the electronic service with Suomi.fi e-Identification. Information on logging in can be found on the Suomi.fi website. You can apply for the mandates required for the electronic service by following the instructions on the Suomi.fi website. The following mandate codes for using the electronic service can be selected in the Suomi.fi service:
- Submitting food notifications
- this mandate allows the assignee to browse and submit notifications related to food products and modify their details on behalf of the assignor.
- Browsing food notifications
- this mandate allows the assignee to submit notifications related to food products on behalf of the assignor and browse the details in notifications that the assignee has submitted.
More detailed instructions on submitting notifications and using the electronic service can be found on the Finnish Food Authority website.
Once the notification has been submitted successfully, the service will send an automatic message of the receipt of the notification to the email address given in the notification. The service will also send information on the notification to the food control authority of the municipality within which the operator submitting the notification is located for information and control purposes. If necessary, the Finnish Food Authority will also send information on notifications to other control authorities (Customs and Fimea).
If the food business operator cannot submit the notification via the electronic service, the food supplement notification can be submitted using a notification form, which can be found, if necessary, on the Finnish Food Authority website.
NOTE: When receiving a notification, the Finnish Food Authority does not assess the legality of the food supplement composition or labelling. Receipt of the notification does not mean that the Finnish Food Authority has approved the notified food supplement as being compliant with the requirements laid down in legislation.
The food business operator is responsible for ensuring that the product complies with the Decree on food supplements and other food legislation. This also requires the operator to perform own-check as they are obligated to do under the Food Act.
A fee is charged for the receipt of the food supplement notification in accordance with the Ministry of Agriculture and Forestry decree on fees payable by the Finnish Food Authority. The current price list can be found on the Finnish Food Authority website.
The invoice for the collection of the fee is sent to the address given by the operator in the notification after its processing.
7 Control
7.1 Own-check
Own-check in accordance with section 15 of the Food Act obligates a food business operator to have in place a system whereby the operator identifies and manages the hazards associated with its operations and ensures that the operations meet the requirements imposed under food legislation. The food business operators must have sufficient and correct information on the food supplements they produce, process, manufacture, import, export, package, market, serve or distribute as food.
In addition to the food product itself, own-check focuses on controlling the conditions of its manufacture, processing, transport, storage and marketing to the extent that the operator is involved in the chain.
In the manufacture and marketing of food supplements, every operator in the chain is responsible for their own operation. Information on the ingredients used must be traceable from raw materials to the final product. Operators must be prepared to trace a defect found in a food supplement upstream in the production chain. In the event a product has to be withdrawn, the destination where defective products have been distributed must be known.
Own-check consists of identification, monitoring and written documentation of risks and so-called critical control points. Own-check also includes an up-to-date plan for action in case a food supplement proves to be in violation of regulations.
The conformity of products can be monitored in the own-check procedure through certificates obtained from suppliers of raw materials and through verification of the accuracy of the recipe. The conformity of labelling must also be checked. In some cases own-check samples must be taken and examined. Where necessary, analyses are conducted to monitor, for example, the microbiological quality of raw materials and products, the composition of food supplements (e.g. vitamin and mineral levels) and the amounts of other characteristic substances or the presence of GMO substances.
Additional information on own-check can be found on the Finnish Food Authority website.
7.1.1 Product defects and withdrawals
If a food business operator considers or has reason to believe, for example as a result of own-check or a consumer complaint (e.g. adverse effect report), that a food the operator has imported, produced, processed, manufactured or distributed is not in conformity with food safety requirements or poses a health hazard, the operator must immediately take action to investigate the matter and contact the food control authority (in practice the health inspector) of its own municipality. Suspicion of the safety or compliance of a food supplement may also come to the operator through the authority. (Figure 4)

Figure 4. An example of action in the event a product defect is identified
If a defect is suspected, the operator must immediately launch a risk assessment process to assess its nature and severity. If there is a suspected adverse effect related to a food supplement placed on the market, the operator must assess the possible link between the food supplement and the adverse reaction as well as the required action to eliminate the adverse reaction and to prevent recurrence of similar adverse effects. Municipal food control authorities will evaluate the adequacy of the operator's risk assessment process and, if necessary, direct the actions taken. The risk assessment process must be part of the operator’s own-check.
If a food supplement is found to be harmful to health and thus does not meet the food safety requirements, the food business operator must immediately initiate procedures to withdraw the food supplement from the market and notify the food control authorities (withdrawal notification submitted to the municipal authorities and to the Finnish Food Authority). The information to be submitted to the authorities must include information about the defective product, the nature of the defect, the traceability of the product (where the product has been acquired and where it has been delivered), the number of defective products and actions that have already been implemented. It is important that all the applicable points of the withdrawal notification are filled in accurately and carefully.
If the product has reached the consumer, the food business operator must also inform the consumer about the recall. (Article 9 of the General Food Regulation). The operator must announce the reason for the recall using effective and appropriate means of communication and, if necessary, instruct the consumers on how to return the defective product.
The recall plan must be part of the operator’s own-check.
Additional information on withdrawals and operator obligations can be found on the Finnish Food Authority website.
Own-check tips for operators manufacturing food supplements, importing food supplements or having food supplements manufactured
Ensure that:
- only authorised vitamins, minerals and compounds of them are used in the food supplement (see Section 3.1)
- the microbiological quality of raw materials and finished food supplements is good
- raw materials do not contain hormones or doping substances or any other prohibited substances (see Section 3.12)
- only authorised additives, flavourings and enzymes are used and their conditions for use are met (see Section 3.4)
- the history of use of the food supplement ingredients as food prior to May 1997 has been verified and the food supplement does not contain novel food ingredients that are not authorised in EU (see Section 3.5)
- the authorised novel food ingredients contained in the food supplement meet the conditions of use set for them (see section 3.5)
- the ingredients contained in the food supplement do not make it a medicinal product. If the food supplement contains substances included on the list of medicines, the operator is advised to apply for a classification from Fimea (see Section 3.7)
- food supplements and their raw materials do not contain contaminants exceeding the permitted maximum amounts (see Section 3.10)
- the raw materials of food supplements do not contain pesticide residues in excess of the permitted maximum amounts (see Section 3.11)
- the safety and suitability of packaging materials and other food contact materials has been verified
- labelling matches the composition
- labelling is correctly executed (see Chapter 4) and the mandatory food information is also correctly provided in distance selling
- any irradiation of dried spice herbs, spices and spice plants is indicated in the labelling in accordance with applicable regulations (see Section 4.4)
- NOTE: The irradiation of other ingredients is not permitted (Decree 852/2000 of the Ministry of Trade and Industry on treatment of foodstuffs with ionizing irradiation)
- appropriate information is provided in labelling on genetically modified ingredients (see Section 4.4)
- food supplements marketed as organic products (organic, bio, eco, etc.) meet the criteria set out in the Regulation on organic products and the labelling presents appropriate information (see Section 4.3)
- the necessary restrictions on use and warnings are shown, taking potential risk groups into account
- NOTE: In the absence of restrictions on use in the information provided on the food supplement, the food supplement may be expected to be safe for all healthy consumers.
- marketing of food supplement is in accordance with regulations and good practice (see Section 4.5)
- the food supplement notification has been submitted to the Finnish Food Authority, it is up-to-date and available for presentation on request (see Section 6.2)
- the traceability of food supplements can be verified on the basis of documents (e.g. supplier’s shipping lists, invoices or other stock records). The food supplements sold must also be traceable if they have been delivered other than directly to the consumer.
Own-check tips for operators distributing food supplements
As an operator selling food supplements, check from your supplier that:
- the food supplements have been notified to the Finnish Food Authority (see Section 6.2)
- the labelling on food supplements is presented in Finnish and Swedish
- the labelling information is legible and easily visible
- the labelling of food supplements marketed as organic products (organic, bio, eco, etc.) indicates that the product is subject to organic control, i.e. the code number of the organic control body (see Section 4.4)
- no medicinal claims are made in the labelling of the food supplement or in its marketing and the claims made on the product are truthful (see Section 4.5)
- the supplier and the food supplement are traceable. However, if no Finnish food business operator can be found in the product labelling, the seller of the product must know the distributor responsible for the product. According to the EU General Food Law (178/2002), the responsibility for the product and its traceability is transferred from one operator to another in the product chain.
The condition of the products should be inspected when they are received. Own-check also includes the monitoring of, for example, shelf life and correct storage temperatures.
7.2 Regulatory control
Food supplements are primarily controlled by the municipal food control authorities, on whose territory the food supplement companies are located. Customs controls food supplements imported from third countries and, in addition, imports from EU countries as a random market surveillance. The border control unit of the Finnish Food Authority controls the import of food supplements of animal origin from third countries and the import of raw materials of animal origin for their manufacture. The Finnish Food Authority directs control operations throughout Finland.
7.2.1 Municipal control authorities
Municipal food control authorities enforce food control and, if necessary, provide advice to food business operators. With regard to food supplements, control focuses primarily on manufacturers, contract manufacturers, importers and wholesalers. Control of the municipal food control authorities mainly focuses on monitoring the function of the food business operator’s own-check (see section 7.1). The product-specific requirements for food supplements are controlled in accordance with the Oiva evaluation guidelines. The special requirements for food supplements are controlled in accordance with Oiva evaluation guideline 12.1, but other Oiva evaluation guidelines are also applied to food supplements.
7.2.1.1 Registration of a food business
Activities related to food supplements, such as manufacture, contract manufacture, import, export and sales, are food-related activities that must be notified to the food control authority of one’s own municipality. The municipal food control authorities receive and process food business notifications (see section 6.1), save the information on the food business registration in the centralized information management system for environmental health care and notify the operator that their information has been saved. Once the data has been saved, the food business is subject to systematic food control and the frequency of inspections is determined for the business based on its risk classification.
7.2.1.2 First inspection
The municipal food control authorities conduct the first inspection on the food business that is the subject of the notification. The inspection can be conducted in the manner considered best by the municipality, either physically or remotely. In connection with the inspection, the municipality provides the food business operator with instructions and advice on legislative requirements, if necessary.
7.2.1.3 Control
After the first inspection, the food business operator is usually subjected to regular controls, whose frequency is based on the risk classification (in Finnish and Swedish only) and Oiva evaluation guidelines, but not in all cases.
The manufacture, contract manufacture and import and/or export of food supplements (EU and third countries) are subject to systematic control and the food business is controlled regularly.
The sale, marketing or other distribution of food supplements is controlled
- regularly if the total turnover of food sales exceeds EUR 20,000 per financial year.
- as needed, if the sales of food supplements are small in scale, i.e. the total turnover of food sales is less than EUR 20,000 per financial year.
If an operator selling food supplements also engages in other food business activities, the turnover of the entire food business is taken into account in the turnover.
However, the activities may be subjected to regular control if, for example, suspected misconduct, whistleblowing or consumer complaints received give rise to suspicion of illegal activities.
Once the control authority is satisfied that the activities are in compliance with the legislation, the food business can again be left outside of regular food controls.
7.2.1.4 Control fees
A basic control fee of EUR 150 is charged annually to food business operators subject to systematic food control. An exception to this is the very small-scale marketing of 1-2 food supplements, in which the total turnover of food sales is less than EUR 20,000 per financial year. The very small-scale marketing of food supplements is not subject to a basic control fee.
In addition to the basic control fee, a fee is charged for the registration of food business operations, the first inspection and other regular food control inspections. Inspections outside regular food controls (e.g. those conducted in connection with suspected misconduct, whistleblowing or consumer complaints) may also be subject to a fee. The fees are determined according to the rates set by the municipality.
Additional instructions and information on control authorities can be found on the Finnish Food Authority (in Finnish and Swedish only).
7.2.2 Customs
Finnish Customs has a nationwide sampling and control network in place for the control of imported food. The Product Safety Unit of the Customs is in charge of regulatory operations and the Customs Laboratory conducts the analyses.
The responsibility for the control of food supplements manufactured within the internal market of EU rests primarily with the control authorities of the country of manufacture. However, according to EU food control regulations, samples may be taken at all stages of production, distribution and marketing. The purpose of this random sampling is to verify that control is effective in the country of origin and the required labelling information is provided on food supplements also in Finnish and in Swedish.
Customs controls food supplements imported from the internal market through random tests of samples taken in importer warehouses. This control is based on the annual control plan of Customs. Since these are internal market samples, the products are not banned pending the analyses. The holder of the goods receives a report of the analyses conducted. The fee laid down in the Decree of the Ministry of Finance is charged for the analyses. If non-conforming products are found, Customs will ban the marketing of the goods remaining in stock. If there is an obvious health hazard, the goods already on the market will also have to be withdrawn by the operator (see Section 7.1.1). Customs informs the Finnish Food Authority about the matter, and the Finnish Food Authority sends a control request to municipal food control authorities to verify that the operator has made the withdrawal and taken any other necessary measures.
Food supplements are also imported to Finland from countries outside the EU. The control of these products falls within the remit of Customs. Customs conducts an assessment of the need to take samples based on the Customs control plan in connection with the clearance of the goods. The distribution of goods of which samples are taken is discontinued pending the analysis results. When the analyses have been completed, the clearance of goods that are in conformance is resumed. Non-conforming food supplements may, if deemed appropriate and with permission of the control authorities, be returned to the foreign distributor or manufacturer, exported from the country, destroyed under the supervision of Customs or modified in order to comply with regulations. The fee laid down by the Ministry of Finance is charged for the control. The Finnish Food Authority forwards information on the analyses conducted by Customs to municipalities for the control of own-check performed by the importers of food supplements.
The classification of preparations as medicinal products in other countries may differ significantly from the classifications applied in Finland. In uncertain cases, Customs always consults Fimea for a classification of the product before taking any control measures.
7.2.3 Finnish Food Authority veterinary border control
Products of animal origin imported from outside the EU are subject to veterinary border control. The importer is responsible for ensuring that the import conditions are met. In some cases, food supplements containing ingredients of animal origin also require a veterinary border check. The import requirements for food supplements containing raw materials of animal origin are specified in section 3.3.
The importer shall be obligated to import the consignment through a border control post with a veterinary border inspection post and to arrange for the consignment to be placed under border control. Veterinary border control is subject to a fee. The border veterinarian of the Finnish Food Authority conducts veterinary border control on products of animal origin. The border veterinarian ensures that the consignment satisfies the conditions required for entry into the EU and either accepts or rejects the consignment.
Additional information on veterinary border control can be found on the Finnish Food Authority website.
7.2.4 Finnish Food Authority
The Finnish Food Authority plans and supervises food control also on food supplements through guides and instructions, and by providing information and training.
Food supplement notifications are submitted to the Finnish Food Authority. Food supplement notifications are explained in greater detail in Section 6.2. The Finnish Food Authority does not control the conformity of food supplements when it processes the notifications but forwards the notifications to municipalities for information and control purposes. A summary of the data provided in the notifications is sent to Fimea and Finnish Customs as well as to other authorities, if necessary. The municipalities and Customs control the validity of food supplement notifications in connection with inspection visits and analyses, or by means of projects, if necessary.
The Finnish Food Authority can include a national control project regarding food supplements in its plans and annual control programmes. Such projects may focus on certain types of businesses (such as gyms), certain food supplement categories (such as calcium preparations) or marketing (nutrition and health claims).
The Finnish Food Authority and Customs Laboratory have agreed that the Finnish Food Authority will forward the analysis findings of the Laboratory and any actions taken by Customs to municipalities four times a year for their information.
The Finnish Food Authority acts as an official national contact point
- for the Rapid Alert System for Food and Feed (RASFF) and
- the Administrative Assistance and Cooperation (AAC) Network.
If Customs suspects that non-conforming food supplements have entered the domestic market, the Finnish Food Authority will notify the municipal authorities and request that they take control measures to verify that the operator has taken adequate measures to rectify the defects and, if necessary, has initiated a withdrawal procedure regarding the defective food supplements.
If necessary, the Finnish Food Authority can also participate in inspection visits to the manufacturing facilities of food supplements and to the warehouses of importers or packers together with the municipal authorities.
7.3 Coercive measures and sanctions
Control measures on food supplements are carried out under the Food Act.
Food control authorities may make a request that is less severe measure than an order or prohibition in cases that do not involve an actual health hazard or the intentional provision of misleading information. The request for corrective measures should, however, always be given in writing and a deadline should be set for complying with the request. A temporary prohibition may be used if, for instance, the labelling of the food supplement must be corrected, or a notification of a food supplement has not been made to the Finnish Food Authority despite a request to do so. A periodic penalty payment is imposed in situations, which involve repeated violation of provisions and orders of the control authorities, or when the severity of the situation otherwise warrants a periodic penalty payment to enforce the decision issued. Under the Food Act, municipal food control authorities may request executive assistance from the police, if necessary.
In addition to coercive measures, the Food Act defines the cases in which the municipal food control authorities are under an obligation to request the police to conduct a pretrial investigation. This obligation arises, for example, in cases where the operator has either intentionally or through negligence provided misleading information about a food or its properties, and the act is not minor in nature.
Coercive measures are explained in greater detail in the Finnish Food Authority guide on the use of administrative coercive measures in food control under the Food Act (in Finnish and Swedish only).
Entry into force
This guideline is valid effective 1 November 2025 and replaces the previous version (Food Safety Authority Evira guideline 17012/5, published on 25 January 2016).
Updates to version 6
- Guideline changed from the Evira guideline to the Finnish Food Authority guideline in an accessible format.
- Legislation and instructions updated.
- Interpretations of interface products added.
- Some labelling examples removed.
- Table for Indicative examples of action taken by control authorities in various situations deleted.
- List in the section Legislation and Norms, with a link to the list of legislation and norms compiled on the Finnish Food Authority website, replaced.
- List of Useful links (links embedded in the guide text for each matter) delete.